Nature’s Boarding Pass: How Proteins Enter Mitochondria
- Details
- Published on 01 September 2025

Scientists Reveal Exciting New Rules for Mitochondrial Protein Delivery.
Caltech researchers have discovered a whole new layer in how proteins get to mitochondria, the energy centers of our cells. This finding challenges long-held beliefs and offers a glimpse into nature’s clever engineering.
For decades, scientists believed that mitochondrial proteins were made completely before being delivered. But the new study, published in Cell, shows that nearly 20 percent of these proteins begin their journey while still being made by ribosomes. This early delivery is called cotranslational import.
What makes this pathway special? The proteins that use it are usually large, complex, multi‑domain molecules, the kind that are difficult to fold correctly if fully formed out in the cell fluid. Folding them early and sending them off right away helps prevent them from getting stuck or forming tangled knots that could clog the mitochondrial gates.
But how does the cell know which proteins need this special route? It turns out there’s a two step signal system at play:
- A mitochondrial targeting sequence (think of it like a boarding pass).
- The appearance of a large folded domain the ‘code’ that unlocks the suitcase so the boarding pass can be used.
As Zikun Zhu, the lead author, puts it: “It’s like having your boarding pass locked in a suitcase. The targeting sequence is the boarding pass, but to access it you need the code to open the suitcase. In this case, the large domain is that code.”
In fact, when the researchers added one of these large domains to proteins usually imported later, they redirected them to the cotranslational pathway showing just how powerful this signal really is.
Unlike cotranslational targeting in other cell parts, mitochondrial targeting happens later only after the big domain is ready. This strategy ensures only the right proteins enter at the right time.
Looking ahead, the team plans to dive deeper into how this system works at a molecular level with hopes of one day tuning it for medical or biotechnological applications.
The topic will be further discussed at the World Mitochondria Society (WMS) annual meeting in Berlin, October 2025, where leading experts will explore how such discoveries are shaping the future of mitochondrial medicine.
Mitochondria: Not Just the Powerhouse, but a Pathogen Defense Hub
- Details
- Published on 27 August 2025
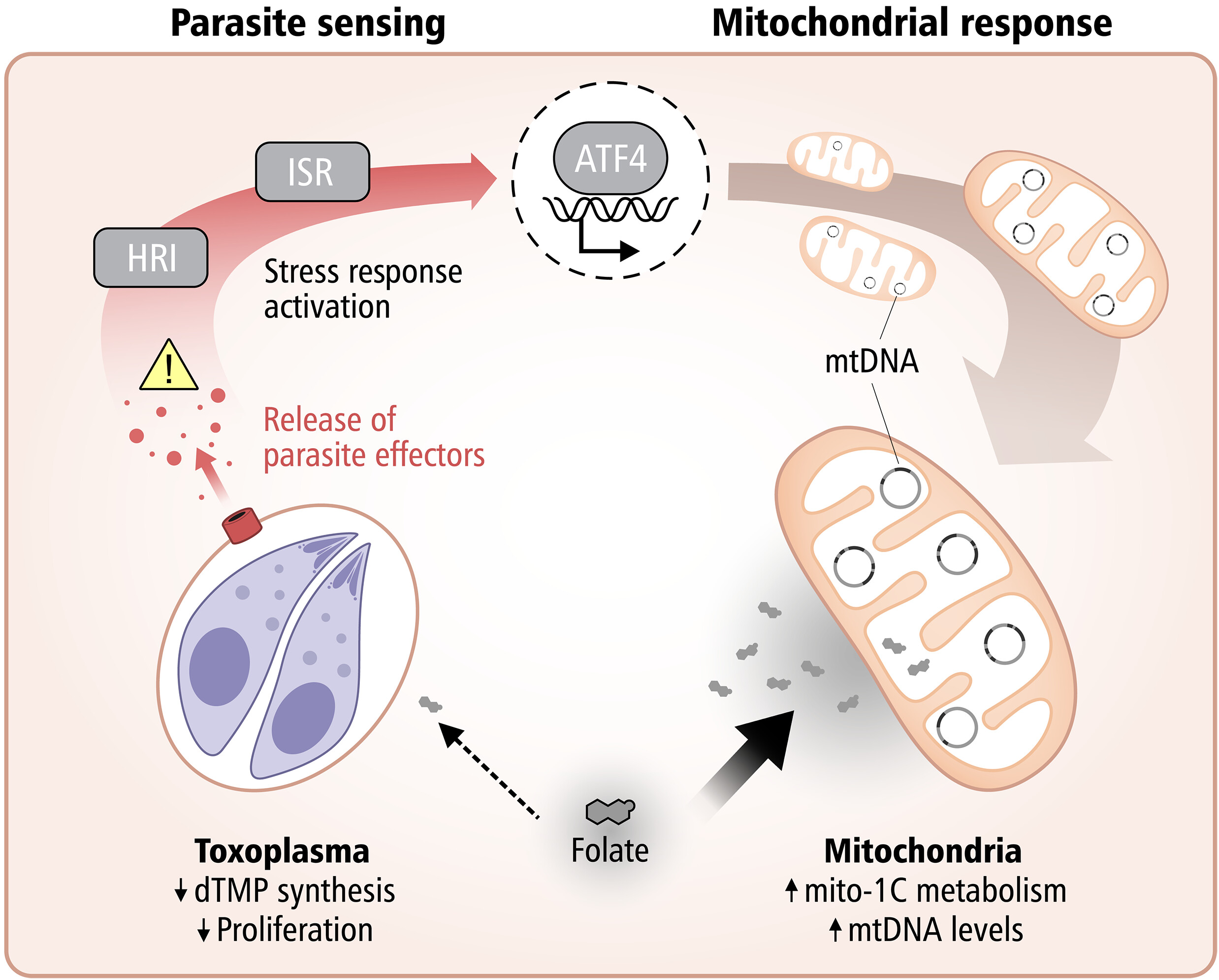
A fascinating new study published in Science from Lena Pernas’s group at the University of California Los Angeles reveals how our cells can starve intracellular parasites, by harnessing their own mitochondria to compete for essential nutrients.
The research demonstrates that mitochondria take on an active defensive role against the parasite Toxoplasma gondii by competing for folate, a vitamin critical for the parasite’s DNA synthesis.
When host cells detect infection, they trigger the integrated stress response, activating ATF4, which boosts mitochondrial one-carbon metabolism. This metabolic shift depletes host folate, effectively starving the parasite and limiting its replication.
This work highlights a striking example of metabolic immunity, where cells weaponize their own metabolic machinery to fight off infection.
How Mitochondria Can Be Matched to Pathology
- Details
- Published on 01 July 2025
From Powerhouse to Precision Tool in Regenerative Medicine
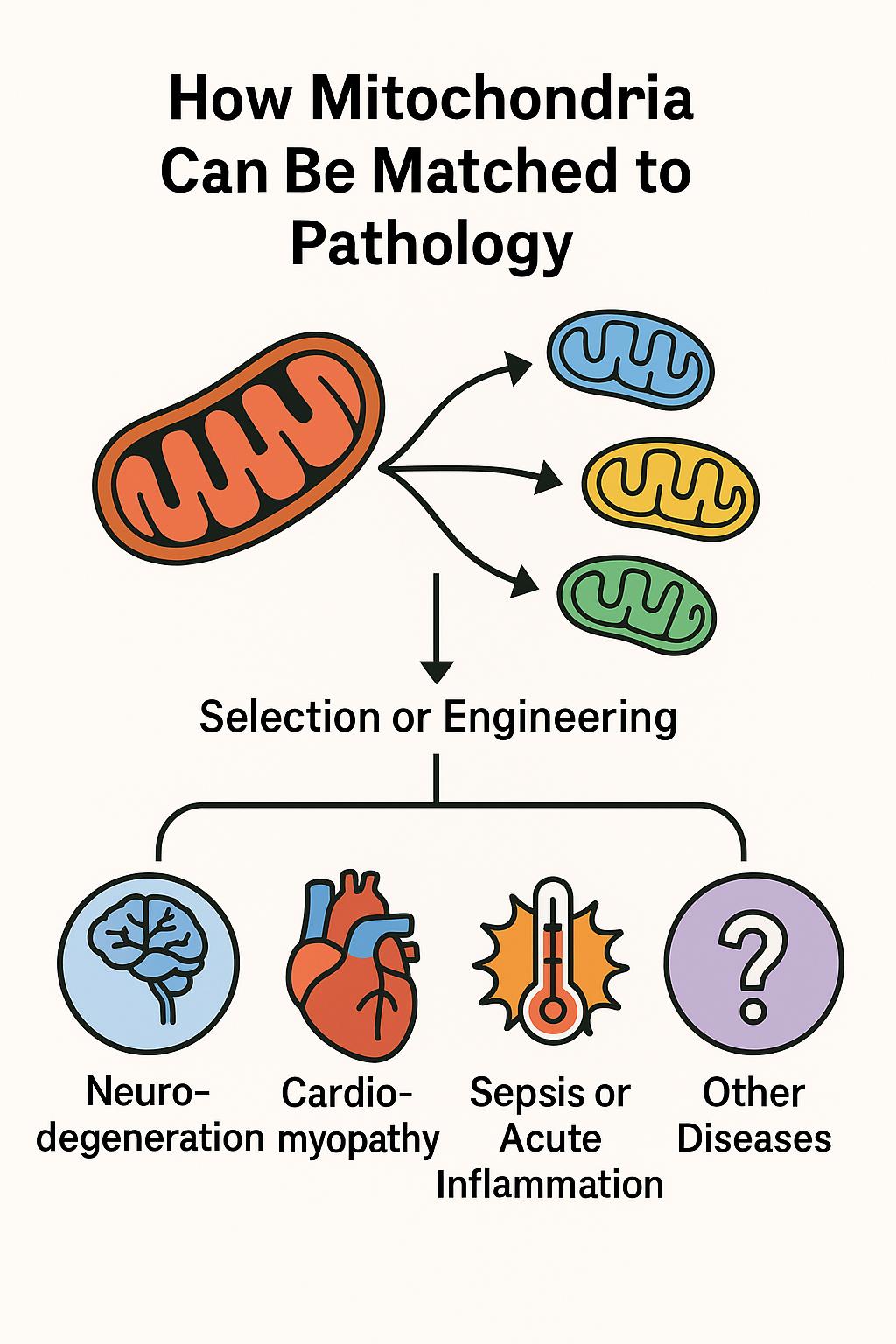
Mitochondrial transplantation is rapidly emerging as a groundbreaking strategy in precision medicine. A recent study published in Cell Death & Disease demonstrates that mitochondria sourced from different cell types and species can exert distinct therapeutic effects, depending on the nature of cellular stress or damage—supporting the concept of pathology-matched mitochondrial therapy.
Led by Xiaomeng Lu the study demonstrates that mitochondria sourced from diverse species (canine, porcine, bovine, etc.) can be safely transplanted into stressed immune and cardiac cells, and even into live animal models, without triggering an immune response. More importantly, these transplanted mitochondria conferred distinct, context-dependent protective effects based on their origin—suggesting a future of tailored mitochondrial therapies.
This approach, referred to as adaptive bioenhancement, proposes that donor mitochondria can be selected or engineered to match the metabolic or immunological needs of specific diseases. The implications are profound: mitochondria are no longer seen merely as cellular batteries, but as programmable organelles capable of restoring balance in a targeted fashion.
Key Concepts Illustrated
• Cross-species delivery is safe and effective: The study found that mitochondria from various mammalian cells were well tolerated in both cell cultures and live mice—even across species barriers.
• Disease-specific matching is possible:
• Neurodegeneration: Mitochondria rich in antioxidant and synaptic-support functions.
• Cardiomyopathy: High-energy, calcium-buffering mitochondria.
• Sepsis & inflammation: ROS-scavenging, immunomodulatory mitochondria.
• Designer mitochondria: Fusion of mitochondrial populations (e.g. HL1 + H9C2) enhanced key proteins (Opa1, Drp1, Mfn1), enabling mitochondrial performance to be optimized for specific stress contexts.
In Vivo Confirmation
In a mouse model of LPS-induced acute inflammation, tail-vein injection of wild-derived mitochondria:
• Boosted grip strength and endurance
• Reduced IL‑6, TNF‑α, and IL‑10 serum levels
• Normalized liver and kidney function markers
• Reduced tissue ROS and lipid peroxidation
Global Perspective – Berlin 2025
This emerging concept of pathology-matched mitochondrial therapy will be at the forefront of the World Mitochondria Society (WMS) Annual Meeting, to be held in Berlin this October.
WMS continues to serve as the global platform for shaping the future of mitochondrial research and therapeutic innovation.
Reference
Xiaomeng Lu - Mitochondrial transplantation: adaptive bio-enhancement. Cell Death & Disease. 2025;15(7):309. https://doi.org/10.1038/s41419-025-07643-8
Mitochondria May Be the Brain’s Secret Sleep Switch
- Details
- Published on 18 July 2025
By World Mitochondria Society
Why do we feel so tired when we don’t sleep?
A new study published in Nature suggests the answer may lie deep inside our brain cells, specifically in their mitochondria, the tiny energy-producing structures often called the cell’s “powerhouses.”
Researchers from the University of Oxford found that when fruit flies were sleep-deprived, a specific group of brain cells showed dramatic changes in their mitochondria. These neurons, which help control the urge to sleep, responded to sleep loss by ramping up energy production and reorganizing their internal machinery.
Mitochondria and Sleep Pressure
They discovered that sleep pressure, what makes us feel sleepy, builds up when mitochondria in key neurons become overworked.
After rest, these changes reversed, suggesting that mitochondria help the brain recover. Even more striking, the scientists were able to dial sleepiness up or down by changing how the mitochondria worked.
When the mitochondria were forced to split into smaller fragments, the flies slept less. When mitochondria were fused into longer structures, the flies felt a stronger need to sleep.
This discovery adds a surprising new piece to the puzzle of sleep science. Until now, sleep was mostly studied as a brain-wide process involving electrical signals or chemical messengers. But this study shows that the buildup of sleep pressure might begin with stress inside individual brain cells.
What’s next?
This research could one day help scientists design treatments for sleep disorders by targeting mitochondrial health. It also opens the door to rethinking sleep not just as a brain state, but as a metabolic response to cellular energy needs.
A new study published in Nature by Gero Miesenböch : https://www.nature.com/articles/s41586-025-09261-y
Cancer’s Energy Hack: Tumor Cells Steal Mitochondria from Nerves to Fuel Metastasis
- Details
- Published on 26 June 2025
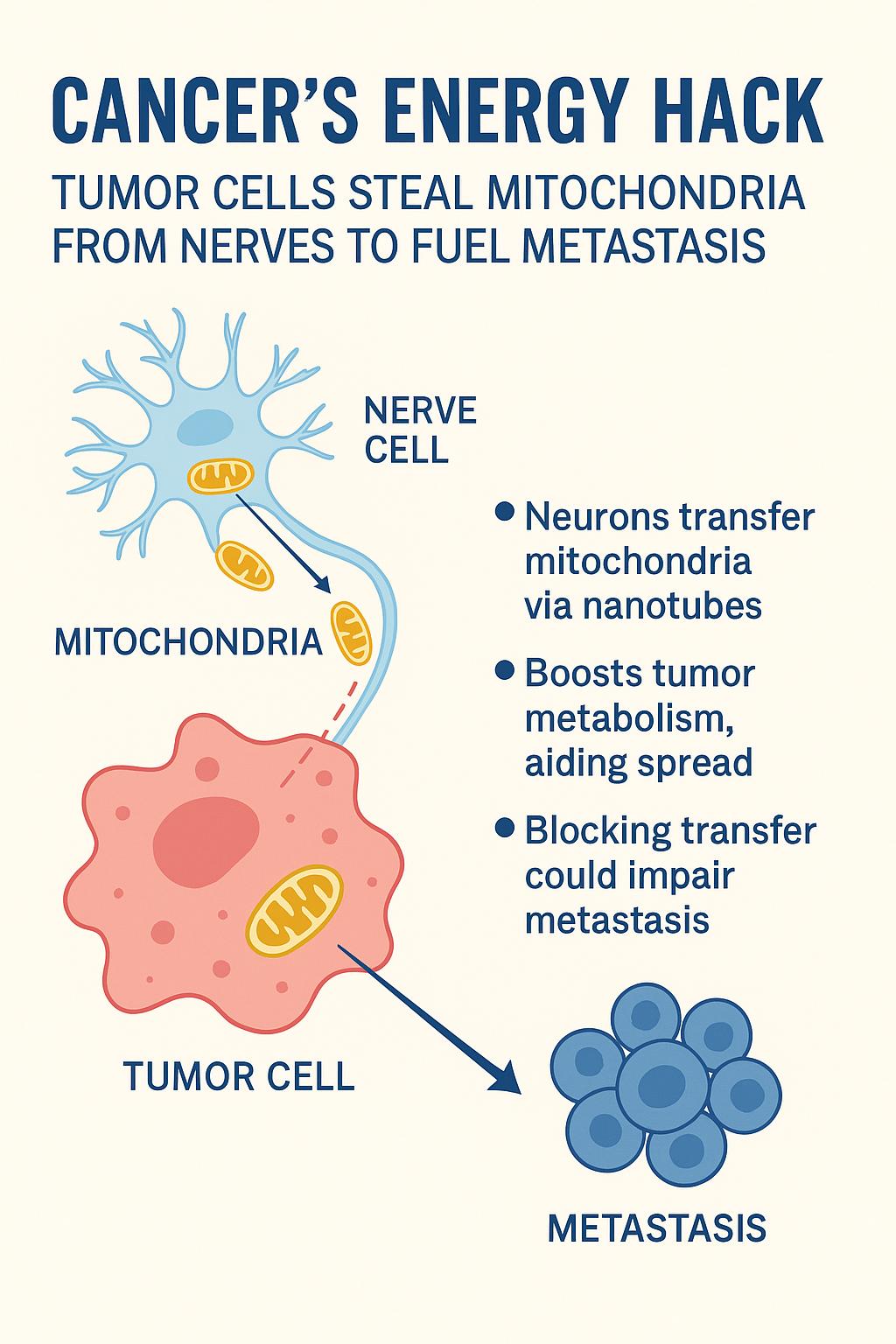
In a striking new study published in Nature, researchers reveal that cancer cells can directly siphon mitochondria from nearby nerve cells — gaining a powerful metabolic edge that helps them spread throughout the body.
A surprising neural alliance
It’s long been known that nerves infiltrate tumors and somehow enhance cancer progression. But the exact mechanism was unclear. This new research, led by Dr. Simon Grelet, shows that neurons surrounding tumors actively produce extra mitochondria and transfer them to cancer cells via nanotubes — thin, tube-like cellular extensions.
These donated mitochondria aren’t damaged or dysfunctional; they’re fully operational, supercharging the cancer cells’ metabolism and helping them survive and proliferate under stress.
A new tool: the MitoTRACER
To track this process, the team developed a genetic sensor called MitoTRACER, which irreversibly labels cancer cells that have absorbed mitochondria from neurons.
In mouse models of breast cancer, these mitochondria-receiving cells were vastly overrepresented in metastatic sites — clear evidence that neuronal mitochondria play a critical role in cancer spread.
Confirmed in human tumors
The findings were validated in human prostate cancer tissue samples. Areas with high nerve density showed more mitochondrial content in nearby cancer cells. Moreover, chemically disrupting the neural supply using botulinum toxin (BoNT/A) reduced the mitochondrial transfer and impaired cancer aggressiveness.
Why it matters :
• A new metabolic weapon: Cancer cells aren’t just reprogramming their own metabolism — they’re outsourcing energy production by stealing power plants from neurons.
• Direct link to metastasis: The mitochondrial boost gives cancer cells the fuel they need to survive oxidative stress and adapt during metastasis.
• A new therapeutic frontier: Targeting the neuron–tumor interaction — by blocking nanotube formation or preventing mitochondrial trafficking — could offer a novel way to slow or stop cancer spread.
More information :
Grelet et al., “Neuronal–tumour cell mitochondrial transfer promotes metastasis,” Nature, 26 June 2025, DOI: 10.1038/s41586-025-09176-8.