How mitochondria organize their DNA to sustain cellular energy
- Details
- Published on 03 April 2026
Researchers from the École Polytechnique Fédérale de Lausanne (EPFL), Switzerland, led by Suliana Manley, and colleagues including Juan C. Landoni, Matthew D. Lycas, Josefa Macuada, Willi Stepp, Roméo Jaccard, Christopher J. Obara, Andrew S. Moore, David Hoffman, Jennifer Lippincott-Schwartz, Wallace Marshall, and Gabriel Sturm.
The work was published in the journal Science on April 2, 2026.
What did researchers observe?
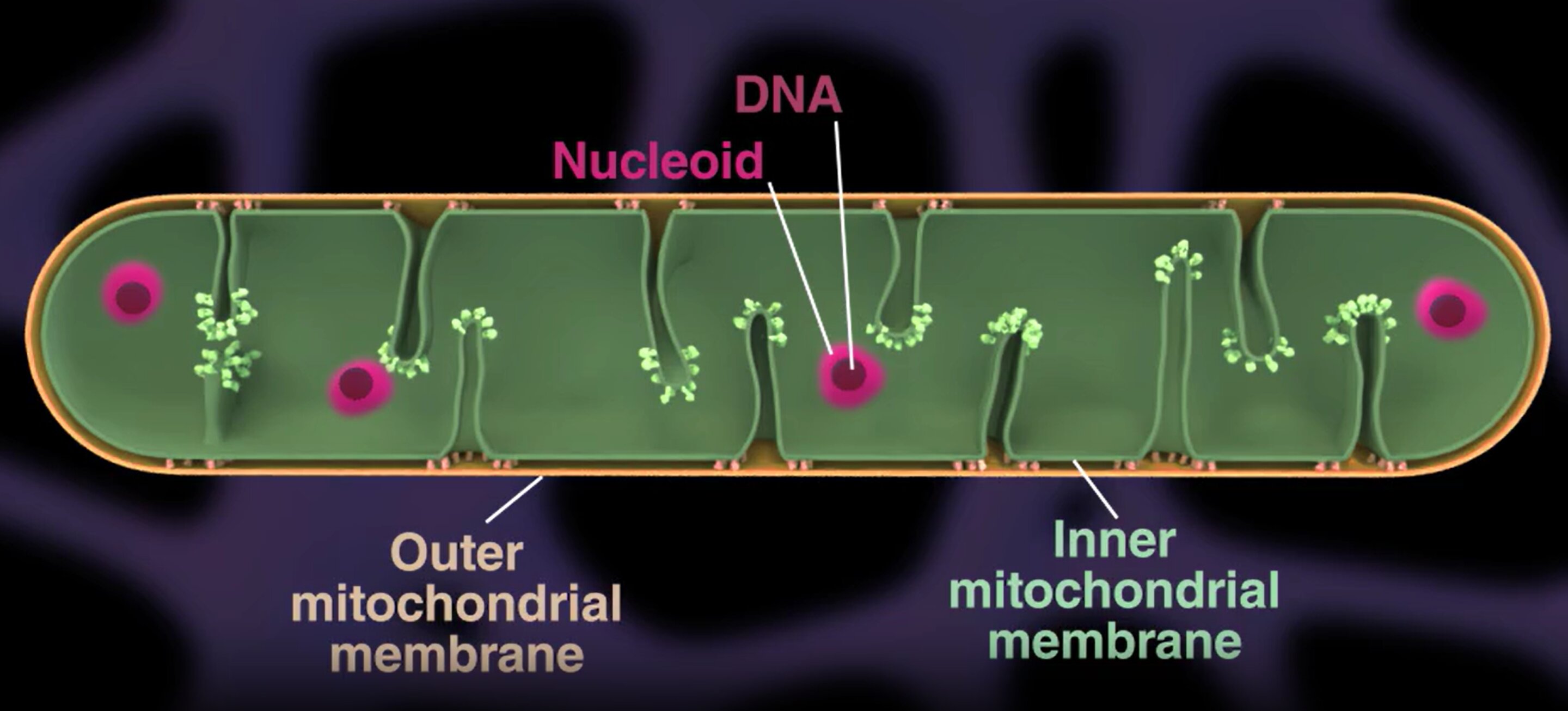
Mitochondria contain small circular DNA molecules grouped into compact structures called nucleoids. These nucleoids are regularly spaced along the mitochondrial network rather than randomly distributed.
Mitochondria contain small DNA clusters called nucleoids. The team found that these nucleoids are evenly spaced along mitochondria, rather than randomly distributed. This spatial organization is actively maintained and is essential for mitochondrial efficiency.
Using advanced live-cell imaging, the scientists showed that mitochondria undergo a physical process called “pearling” , transient bead-like constrictions that help distribute nucleoids regularly along the mitochondrial network.
This ordered distribution ensures:
- Balanced mitochondrial gene expression
- Efficient energy production
- Proper inheritance of mitochondrial DNA during cell division
When this organization is disrupted, mitochondrial function becomes uneven, potentially leading to cellular dysfunction.
A physical mechanism, not just genetics
The study demonstrates that biophysical forces inside mitochondria help control DNA positioning. This means mitochondrial performance depends not only on genetic content, but also on how that DNA is physically organized.
The implications
This mechanism may help explain mitochondrial dysfunction observed in:
- Aging
- Neurodegenerative diseases
- Metabolic disorders
It also opens the possibility of targeting mitochondrial structure - not only mitochondrial genes - as a future therapeutic strategy.
Mitochondria maintain cellular energy by carefully spacing their DNA. This spatial organization is an active, physical process that directly supports mitochondrial function and cellular health.
Reference
Juan C. Landoni et al., Pearling drives mitochondrial DNA nucleoid distribution.Science392,102-109(2026). DOI:10.1126/science.adu5646
Mitochondria Could Help Make Cancer Immunotherapy More Effective
- Details
- Published on 31 March 2026

A new study published in Nature by Ping-Chih Ho from Department of Oncology, University of Lausanne, Lausanne, Switzerland and reported by Drug Target Review suggests that improving the energy systems of immune cells may boost the success of CAR-T cell therapy, an advanced treatment used to fight cancer.
What is the problem?
CAR-T therapy uses a patient’s own immune cells to attack cancer. However, these cells often become “exhausted” after some time. When this happens, they lose their ability to fight tumors effectively.
Where do mitochondria come in?
Mitochondria are the “power plants” of cells. They produce the energy that immune cells need to survive and function.
The study shows that exhausted immune cells have damaged or weak mitochondria. As a result, they cannot maintain their activity against cancer.
What did researchers find?
When scientists restored mitochondrial function, the immune cells:
- Lived longer
- Produced more energy
- Attacked tumors more efficiently
- Stayed active for a longer period
Why this matters
This means that improving mitochondrial health could:
- Make CAR-T therapy stronger
- Help prevent cancer relapse
- Improve long-term treatment success
- Expand this therapy to more cancers
The big idea
Instead of only modifying immune cells to recognize cancer, researchers may also need to boost their energy capacity. Healthy mitochondria could allow these cells to remain active and continue fighting tumors.
Key message
Mitochondria are not just energy producers, they may determine how well cancer immunotherapies work.
By protecting or enhancing mitochondrial function, scientists may improve the effectiveness and durability of next-generation cancer treatments.
Reference
Xu, Y., Shangguan, Y., Chuang, YM. et al. Proteasome-guided haem signalling axis contributes to T cell exhaustion. Nature (2026).
Could fixing mitochondria restore insulin-producing cells?
- Details
- Published on 12 February 2026
Mitochondria - the tiny power plants inside our cells may be a master switch in diabetes, controlling not just energy production but the very identity of key metabolic cells.
Core finding:
University of Michigan researchers lead by EMILY M. WALKER and team show that when mitochondria are damaged in insulin-producing pancreatic β-cells, a specific cellular stress response switches on, causing these cells to become “immature,” produce too little insulin, and essentially stop functioning as true β-cells. Crucially, the team finds that this same mitochondrial stress program can also be triggered in liver and fat cells, hinting at a unifying mechanism behind the multi-organ breakdown seen in diabetes.
How they tested it::
Using mice, the scientists disrupted three separate pillars of mitochondrial health: mitochondrial DNA, the pathway that clears damaged mitochondria, and the system that keeps a healthy pool of mitochondria in the cell. Despite these different hits, each manipulation triggered the exact same stress response and led β-cells to lose maturity and insulin output, revealing a common “danger signal” from mitochondria to the cell nucleus.
Beyond the pancreas:
Because diabetes affects far more than the pancreas, the team repeated the experiments in liver cells and fat-storing cells, again seeing the same stress response and impaired cell maturation and function. Senior author Scott Soleimanpour notes that losing β-cells is the most direct route to type 2 diabetes, but these results suggest a shared mitochondrial glitch across tissues could be the deeper root cause.
A possible way to reverse it:
In a hopeful twist, mitochondrial damage did not kill the cells, raising the possibility that their function could be restored. When the researchers treated mice with ISRIB, a drug that blocks the stress response pathway, β-cells regained their ability to control blood sugar within four weeks, effectively reversing the mitochondrial-driven dysfunction in this model.
What’s next:
The team confirmed key findings in human pancreatic islet cells and is now dissecting the disrupted pathways in more detail, aiming to repeat the rescue in cells from people with diabetes. If successful, targeting this mitochondrial stress signaling could shift diabetes treatment from managing blood sugar to correcting a fundamental cellular error – turning the cell’s “powerhouse” into a therapeutic entry point.
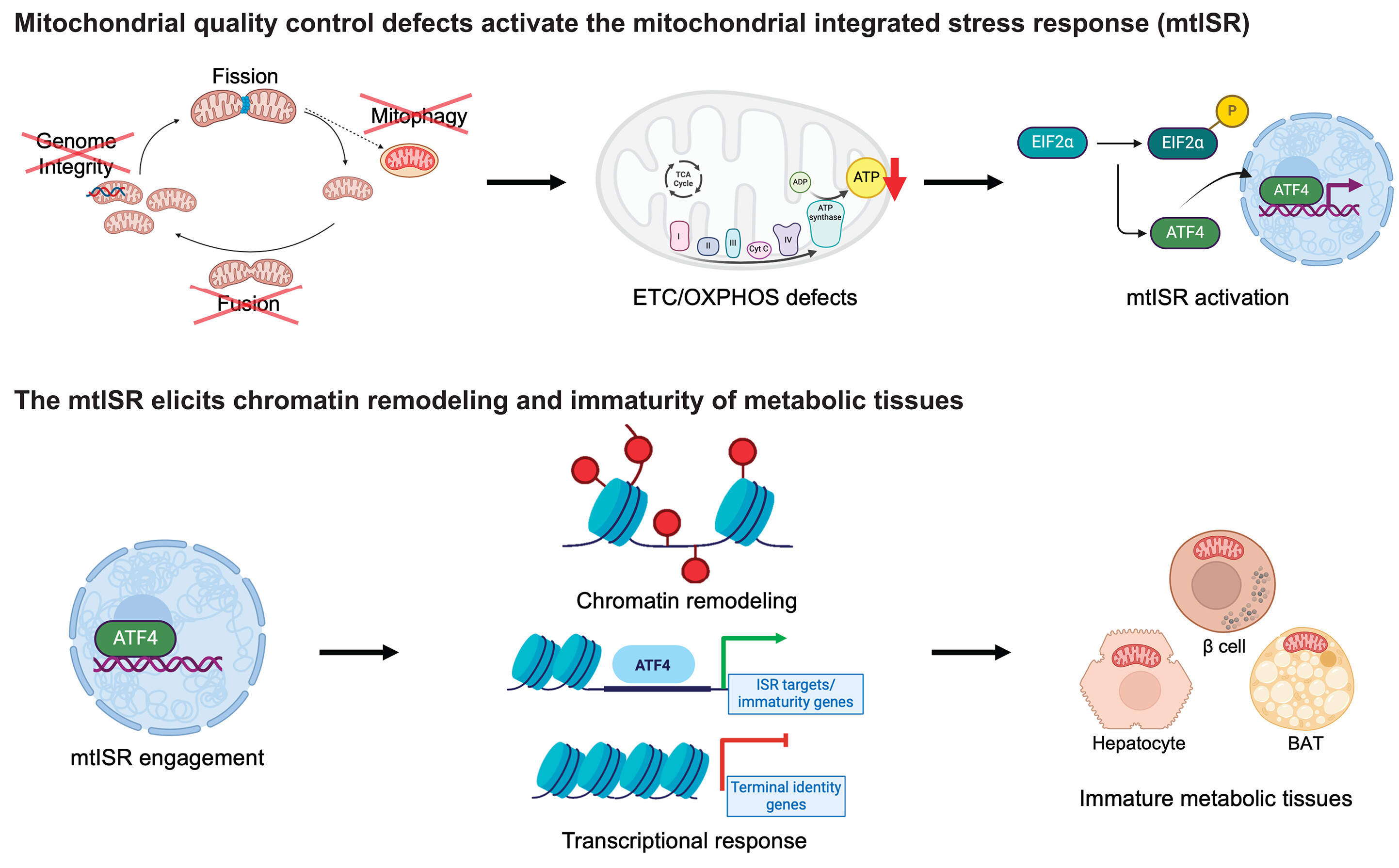
About Image
A retrograde mitochondrial signaling cascade induces the loss of identity and maturity in metabolic tissues.
Numerous defects in the mitochondrial quality control machinery are capable of eliciting defects in the ETC-OXPHOS system, which acts as a retrograde signal to trigger the mtISR. Engagement of the mtISR induces chromatin remodeling and transcriptional and functional immaturity in metabolic tissues. TCA, tricarboxylic acid; ADP, adenosine 5′-diphosphate; ATP, adenosine 5′-triphosphate; BAT, brown adipose tissue. [Figure created with BioRender.com]
Reference
Emily M. Walker et al. ,Retrograde mitochondrial signaling governs the identity and maturity of metabolic tissues.Science388,eadf2034(2025).DOI:10.1126/science.adf2034
Organelle Therapy Encapsulated Mitochondria Restore Function in Parkinson’s and Genetic Disorders: A Breakthrough
- Details
- Published on 23 March 2026
 A new study published in Cell by Xingguo Liu and colleagues demonstrates a major advance in mitochondrial medicine. The researchers show that transplanted encapsulated mitochondria can restore cellular energy and improve disease outcomes in preclinical models. This work introduces a scalable strategy for delivering functional mitochondria to diseased tissues, a key challenge that has limited mitochondrial transplantation.
A new study published in Cell by Xingguo Liu and colleagues demonstrates a major advance in mitochondrial medicine. The researchers show that transplanted encapsulated mitochondria can restore cellular energy and improve disease outcomes in preclinical models. This work introduces a scalable strategy for delivering functional mitochondria to diseased tissues, a key challenge that has limited mitochondrial transplantation.
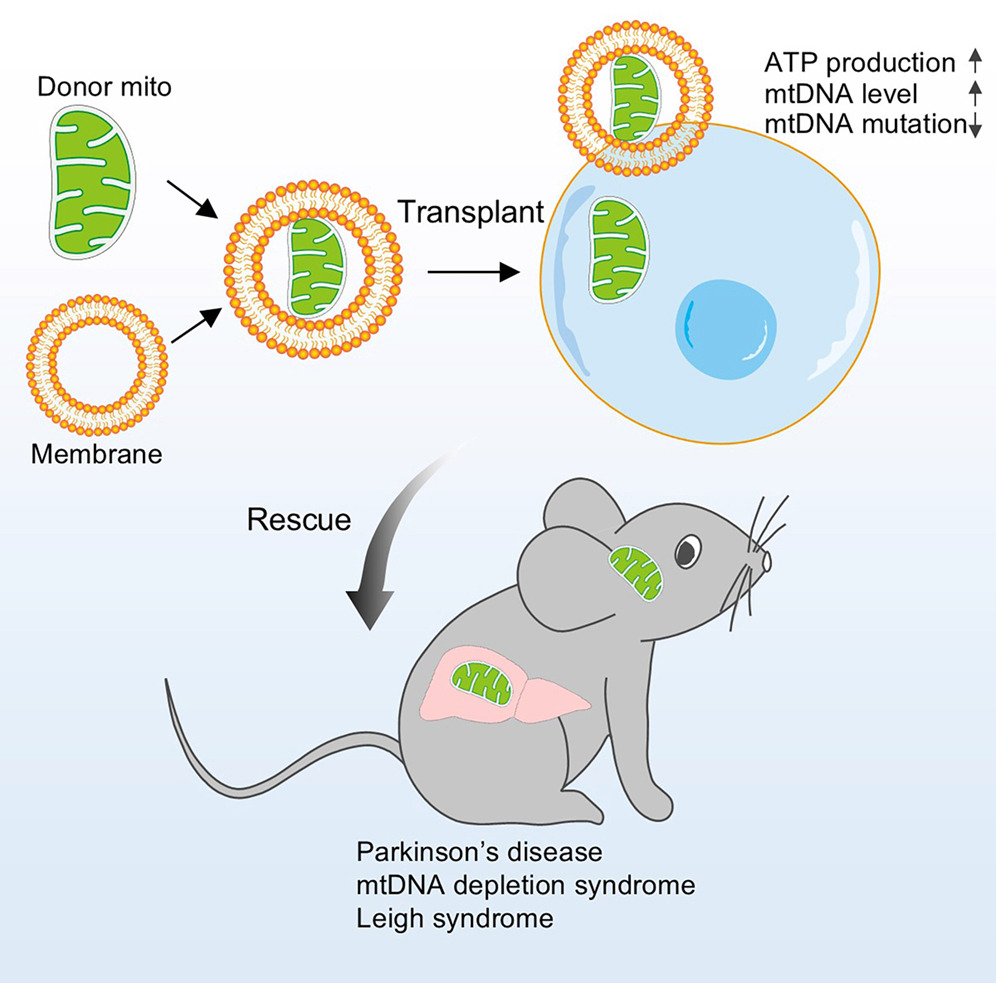
Researchers developed a delivery system in which healthy mitochondria are enclosed within vesicles derived from red blood cell membranes, forming mitochondrial capsules. This encapsulation allowed efficient transfer of mitochondria into cells and tissues in both mice and non human primates.
Once delivered, the transplanted mitochondria integrated with host cells and compensated for mitochondrial DNA defects. The approach restored bioenergetic activity in patient derived cells and corrected biochemical abnormalities associated with mitochondrial disease.
The therapeutic impact extended to in vivo disease models. In mouse models of mitochondrial DNA depletion and Leigh syndrome, treatment improved mitochondrial function and reduced systemic defects. In a Parkinson’s disease model, mitochondrial capsules reduced neuronal loss and improved motor performance.
These findings position mitochondrial transplantation as a potential organelle therapy, a regenerative strategy that replaces dysfunctional cellular components. This approach may open new options for inherited mitochondrial disorders, neurodegenerative diseases, and conditions linked to energy failure.
The study also highlights a conceptual shift. Mitochondria are not only metabolic regulators but transferable functional units that can be engineered and delivered as therapeutics. This supports growing interest in mitochondrial transfer, extracellular vesicle mediated communication, and regenerative bioenergetics as emerging areas in precision medicine.
This breakthrough will be discussed at the annual meeting of the World Mitochondria Society, where experts will explore the clinical potential of mitochondrial transfer and next generation mitochondrial therapies.
Reference
Du S, Long Q, Zhou Y ...
Transplantation of encapsulated mitochondria alleviates dysfunction in mitochondrial and Parkinson’s disease models
Cell, 2026; 0
Is Depression a Cellular Energy Crisis?
- Details
- Published on 12 February 2026

New research suggests that major depressive disorder may involve disruptions in how cells produce and manage energy. Scientists report that mitochondrial function, which controls the production of the energy molecule adenosine triphosphate (ATP), may contribute to the persistent fatigue and low energy commonly experienced in depression.
Depression has traditionally been understood as a disorder involving neurotransmitters, brain circuits, and psychological stress. New research suggests another possibility. Depression may also involve a disturbance in the way cells produce and regulate energy.
Scientists studying young adults with major depressive disorder discovered significant changes in cellular energy metabolism involving adenosine triphosphate, the molecule that fuels nearly every biological process in the body. Mitochondria, the structures inside cells that generate ATP, appear to function differently in individuals with depression.
Using ultra high field brain imaging and metabolic analysis of blood cells, researchers measured ATP levels and ATP production in both the brain and immune cells. The results revealed an unexpected pattern.
Participants with depression showed higher ATP production rates in the brain and elevated ATP levels in immune cells at rest. These increases were strongly associated with the severity of fatigue symptoms.
However, when researchers exposed the cells to metabolic stress, the pattern reversed. Cells from depressed individuals showed a reduced ability to increase ATP production when energy demand increased. This finding indicates impaired mitochondrial flexibility and reduced energetic reserve.
The results suggest that the cellular energy system may already be operating near its limits even when the body is at rest. When the brain requires additional energy for cognitive effort, emotional regulation, or stress responses, the system may struggle to respond.
Researchers describe this as a compensatory phase of the disorder. Early in depression, cells increase ATP production to maintain stable energy levels. Over time, the reduced ability to meet higher energy demands may contribute to fatigue, slowed thinking, and reduced motivation.
Importantly, the same energy signatures were observed in both brain measurements and circulating immune cells. This suggests that depression may involve system wide changes in cellular energy metabolism rather than being limited to brain chemistry alone.
If confirmed in larger studies, these findings could shift how scientists understand depression. Instead of focusing only on neurotransmitter imbalances, researchers may increasingly view depression as a disorder involving mitochondrial function and cellular bioenergetics.
Understanding this process could open new avenues for treatments aimed at restoring mitochondrial health and improving energy metabolism in the brain.
Reference
Cullen, K.R., Tye, S.J., Klimes-Dougan, B. et al. ATP bioenergetics and fatigue in young adults with and without major depression. Transl Psychiatry (2026). https://doi.org/10.1038/s41398-026-03904-y
More Articles...
- Nanoflowers help cells share mitochondria and restore energy
- Cells share mitochondria to protect nerves from pain When cells share mitochondria, nerve pain can be reduced
- A Mitochondrial Protein May Hold the Secret to Longevity
- Sugar first, mitochondria later: how brain immune cells respond to injury