A Chimera that Breaches Mitochondria
- Details
- Published on 20 June 2025

A Chimera that Breaches Mitochondria
A milestone in mitochondrial biology
A research team from the University Medical Center Göttingen has developed a synthetic chimera that can cross both mitochondrial membranes and selectively block mitochondrial protein synthesis—without altering nuclear DNA.
For the first time, scientists can now disable one mitochondrial protein at a time in living cells, enabling real-time study of bioenergetics, redox balance, and disease pathways.
This innovation bypasses long-standing genetic barriers and opens new possibilities for studying and treating mitochondrial dysfunction in aging, neurodegeneration, and metabolic disease.
Mitochondria Take the Lead: How Metabolism Rewrites Cell Fate
- Details
- Published on 19 June 2025
A study led by Scott W. Lowe (Nature, 2025) brings mitochondria into the spotlight — not just as power generators, but as active decision-makers in stem cell fate.
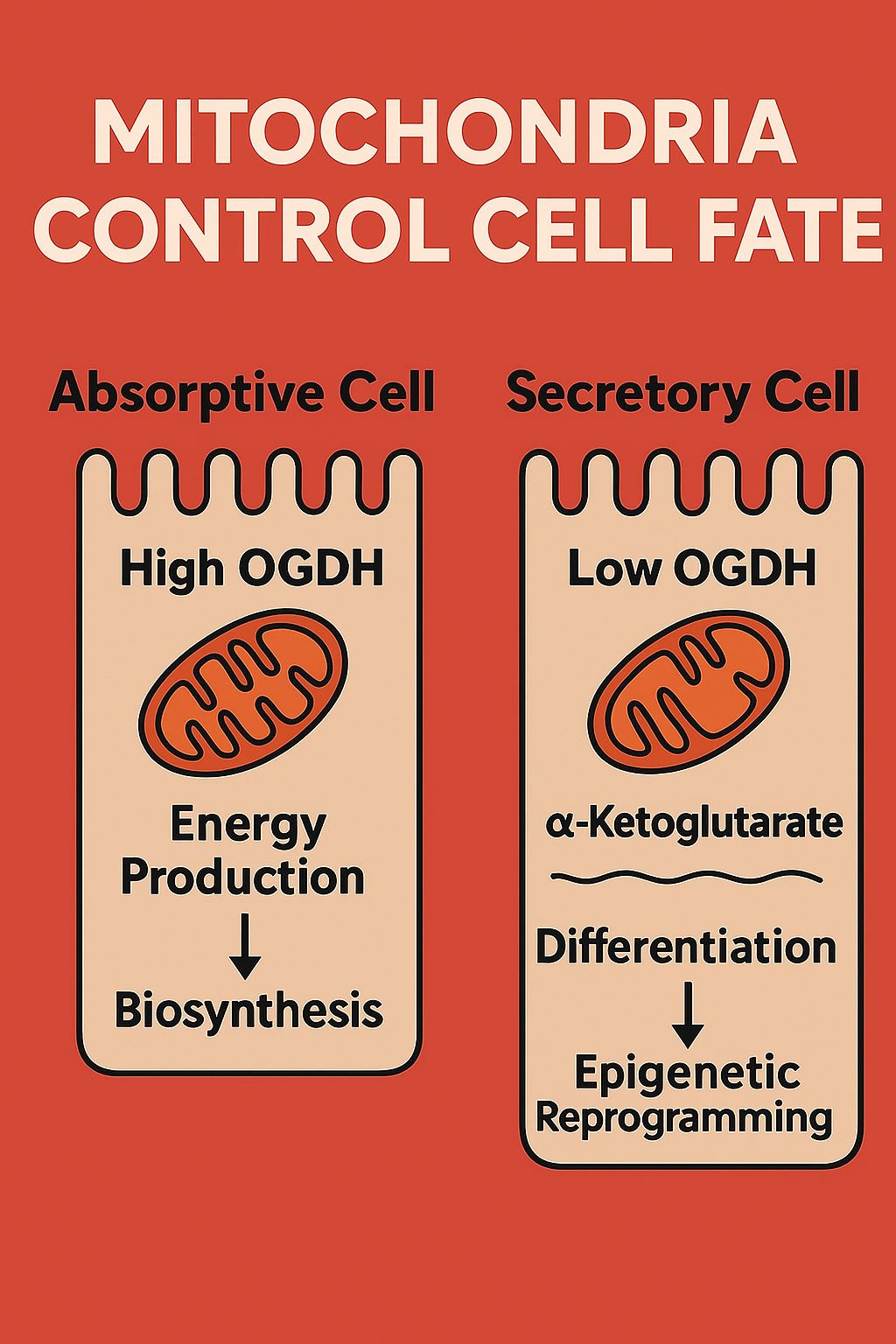
At the heart of the discovery is OGDH, a key enzyme in the mitochondrial TCA cycle. Traditionally seen as a simple metabolic actor, OGDH is now revealed as a master regulator of cell identity in the intestinal epithelium.
- In absorptive cells, high OGDH activity supports energy production and biosynthesis through oxidative phosphorylation, driving cell growth.
- In contrast, low OGDH levels in secretory cells cause an accumulation of α-ketoglutarate (αKG) — a metabolite that acts as a signaling molecule, inducing epigenetic reprogramming and pushing cells toward differentiation.
Using inducible OGDH knockdown in mice, 3D organoid cultures, metabolic tracing, and transcriptomics, the researchers demonstrated that modulating αKG levels can shift stem cell
Mitochondrial Pyruvate Carrier: A Gateway to Future Metabolic Therapies
- Details
- Published on 22 April 2025

Structural and mechanistic insights unveil MPC as a strategic target in cancer, liver disease, and mitochondrial medicine. World Mitochondria Society 2025 Copyright
Fifty years after the mitochondrial pyruvate carrier (MPC) was first identified, researchers have now resolved its molecular structure and mechanism of action. In a landmark study published in Science Advances, Sichrovsky et al. (2025) unveiled how this critical mitochondrial complex mediates pyruvate transport and how its inhibition could be leveraged for therapeutic purposes in cancer, metabolic disorders, and more.
What Is the MPC and Why Is It Important?
The mitochondrial pyruvate carrier (MPC) is a protein complex located in the inner mitochondrial membrane. It enables the import of pyruvate, a central metabolite derived from glycolysis, into mitochondria where it fuels the TCA cycle and ATP production. This process is fundamental to energy metabolism, redox regulation, and biosynthesis.
Until now, the exact structure and working mechanism of the MPC had remained elusive, limiting our ability to design targeted drugs.
Major Discoveries of the Study by Prof. Edmund Kunji and his teams
Molecular Structure of MPC:
The authors used cryo-electron microscopy to capture the architecture of the human MPC complex. They discovered that MPC forms a heterodimeric transport unit (MPC1/MPC2), creating a selective channel that guides pyruvate across the inner mitochondrial membrane.
Mechanism of Transport and Inhibition:
The study revealed how small-molecule inhibitors bind to the MPC complex and block its function, offering a blueprint for drug development. Structural analysis pinpointed specific binding sites that explain both transport dynamics and inhibition sensitivity.
Conserved Functionality:
Evolutionary conservation of the MPC mechanism across species (including yeast and human) underscores its universal biological role in cellular energy homeostasis.
Therapeutic Implications
Cancer:
Some tumors overexpress MPC to fuel high mitochondrial activity. MPC inhibitors could starve these cells of essential metabolites, selectively disrupting their growth.
Metabolic Diseases:
In conditions like non-alcoholic fatty liver disease (NAFLD), blocking MPC forces hepatocytes to burn fat instead of relying on glucose, leading to reduced liver fat accumulation.
Regenerative Medicine & Hair Growth:
MPC inhibition has been shown to stimulate lactate production, which may promote hair follicle cell activation, opening potential new treatments for alopecia.
Mitochondrial Dysfunction & Neurodegeneration:
Targeting MPC may allow modulation of energy metabolism in neurodegenerative and mitochondrial diseases, where ATP production and redox balance are impaired.
Broader Impact
Drug Development:
The structural elucidation of MPC provides a molecular framework for designing selective modulators, setting the stage for new classes of metabolic drugs.
Precision Medicine:
Understanding individual differences in MPC structure/function may lead to personalized metabolic therapies tailored to genetic or disease-specific metabolic profiles.
Synthetic Biology & Bioenergetics:
The detailed MPC model can inform the engineering of customized metabolic pathways, supporting advances in synthetic biology, cell therapies, and biotechnology.
Statement of Prof. Marvin Edeas and Prof. Volkmar Weissig, chairman of World Mitochondria Society, "The study by Sichrovsky et al. marks a strategic breakthrough in the field of mitochondrial biology and cellular metabolism. By revealing the detailed structure and transport mechanism of the mitochondrial pyruvate carrier (MPC), the researchers have clarified how a key metabolic gatekeeper functions at the molecular level. This discovery is important because it gives scientists the precise blueprint needed to design drugs that selectively modulate mitochondrial metabolism. Such interventions could target cancer cells that rely on mitochondrial energy, help treat liver conditions by shifting how cells use nutrients, and even promote regeneration in other tissues."
Overall, this work transforms the MPC from a poorly understood protein into a strategic therapeutic target. It opens new possibilities for developing mitochondria-targeted treatments that are safer, more effective, and tailored to the metabolic needs of each disease.
Prof. Edmund Kunji from the University of Cambridge will give a major talk entitled Targeting mitochondrial pyruvate carrier: impact on future metabolic therapies, during the Targeting Mitochondria 2025 Congress, which will be held on October 22-24, in Berlin Germany.
Reference:
Sichrovsky, M., Lacabanne, D., Ruprecht, J.J., Rana, J.J., et al. (2025). Molecular basis of pyruvate transport and inhibition of the human mitochondrial pyruvate carrier. Science Advances, 18 April 2025. DOI: 10.1126/sciadv.adw1489
Timing of Mitochondrial Dysfunction Found to Shape Lifespan
- Details
- Published on 28 April 2025
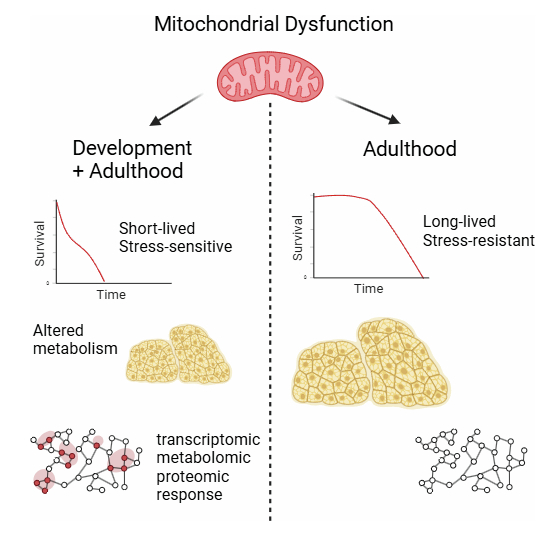
A new study published in EMBO Reports highlights that when mitochondrial complex I (CI) dysfunction occurs dramatically influences lifespan and stress resilience. Using Drosophila models, researchers uncovered striking differences depending on the life stage at which mitochondrial impairment begins:
- Early developmental dysfunction leads to shortened adult lifespan and poor stress resistance.
- Adult-onset dysfunction, despite causing up to a 75% reduction in CI activity, allows flies to live longer and remain stress-resistant.
- Maladaptive biological responses triggered during development—not mere developmental defects—drive the negative outcomes.
- Molecular analyses revealed unique transcriptomic, proteomic, and metabolomic changes in short-lived flies.
The findings suggest that mitochondrial health during early life stages is critical for setting long-term health trajectories. This discovery opens new avenues for developing therapies that target mitochondrial function during key developmental windows to promote healthy aging.
This subject will be talk during the Targeting Mitochondria 2025 congress, which will be held on October 22-24, in Berlin Germany.
Dnmt3a mutation enhances mitochondrial function in blood stem cells, leading to clonal expansion and increased disease risk
- Details
- Published on 17 April 2025

The Jackson Laboratory’s recent study, published on April 16, 2025, in Nature Communications, uncovers how a common age-related mutation in the Dnmt3a gene enhances mitochondrial function in blood stem cells, leading to clonal hematopoiesis—a condition that increases the risk of heart disease, blood cancers, and other illnesses.
Key Findings
- Dnmt3a Mutation and Mitochondrial Enhancement: The Dnmt3a mutation boosts mitochondrial energy production in blood stem cells, granting them a self-renewal advantage.
- Development of Clonal Hematopoiesis: This advantage leads to clonal hematopoiesis, where mutated stem cells dominate, increasing the risk of various diseases.
- Prevalence in Aging Population: Clonal hematopoiesis is common in the elderly, affecting over half of individuals aged 80 and above.
- Inflammatory Impact: Mutated stem cells produce inflammatory molecules that disrupt blood production and weaken the immune system.
- Therapeutic Potential: The study suggests that targeting the enhanced mitochondrial function could be a strategy to prevent or treat clonal hematopoiesis.
These findings show that Dnmt3a-mutated cells rely on enhanced mitochondrial function to dominate,” said senior author Dr. Jennifer Trowbridge. “Targeting this metabolic vulnerability could be a promising strategy to prevent or treat clonal hematopoiesis and its downstream effects.
Marvin Edeas, INSERM, University of Paris, co-organiser of the Targeting Longevity & Mitochondria congress, stated, “This discovery brings new insight into how the aging process is closely linked to energy production in our cells, especially through mitochondria, the cell’s powerhouses. When mitochondria become overactive due to certain mutations, they give some cells an unfair growth advantage, which can lead to harmful conditions like clonal hematopoiesis and increase the risk of diseases such as blood cancer or heart problems. By targeting and calming this mitochondrial overactivity, we may be able to stop these abnormal cells from expanding and protect against age-related diseases. This could represent an important step toward healthier aging.
In a separate publication in Nature published April 16th, Dr. Jennifer Trowbridge provided preclinical rationale for investigating metformin as a preventive intervention against DNMT3A R882 mutation-driven clonal haematopoiesis in humans.
What is DNMT3A?
- It is de novo methyltransferase, meaning it can establish new methylation patterns during development and in adult tissues.
- It plays a crucial role in stem cell differentiation, development, and maintaining genomic stability.
- Mutations in Dnmt3a are commonly found in clonal hematopoiesis, leukemias, and other age-related diseases.
- Recent research shows these mutations can alter mitochondrial function, giving mutant cells a growth advantage.
More Articles...
- Zika Virus NS1 Hijacks Mitochondria Through Tunneling Nanotubes for Immune Evasion
- How Mitochondria Organize Their Powerhouse Machinery for Optimal Performance
- Revolutionary Breakthrough in Mitochondrial Production Offers Hope for Degenerative Diseases
- Why Our Cells Slow Down with Age: The Mitochondria Mystery