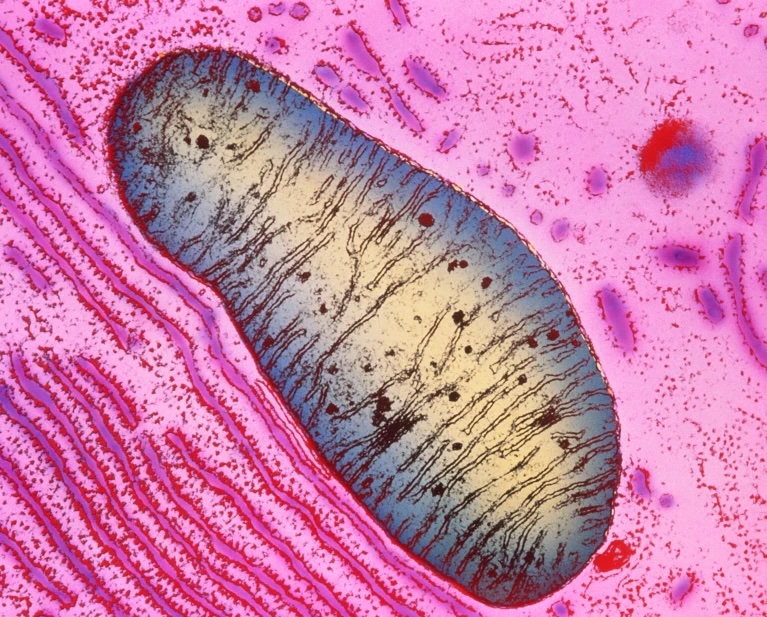
Microglia Transfer Healthy Mitochondria to Rescue Neurons from Neurodegeneration
- Details
- Published on 29 January 2025
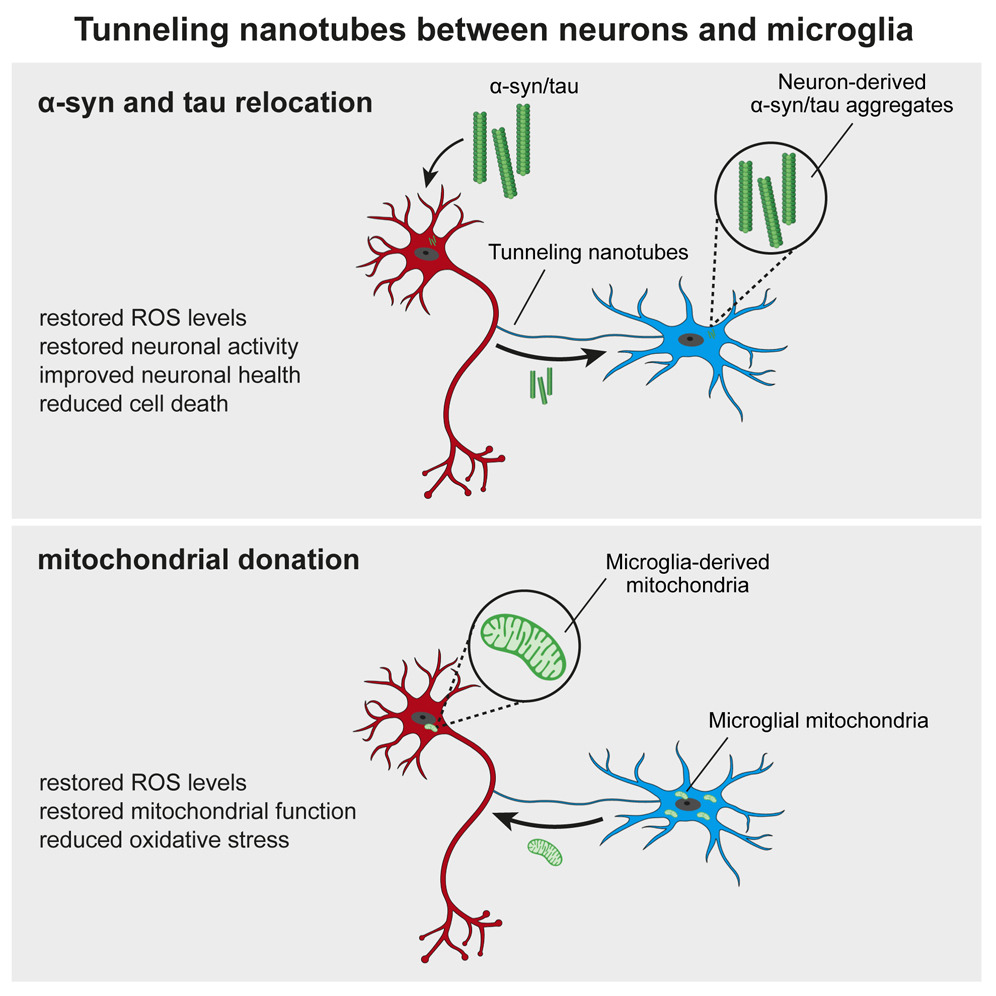
A new study led by Prof. Michael T. Heneka from the Luxembourg Centre for Systems Biomedicine, University of Luxembourg, uncovers a crucial neuroprotective role of microglia in mitochondrial health. Published in Neuron, the research reveals how microglia use tunneling nanotubes (TNTs) to transfer healthy mitochondria to stressed neurons, rescuing them from toxic protein aggregates linked to Parkinson’s and Alzheimer’s diseases.
TNTs act as direct cellular bridges, allowing microglia to exchange organelles, vesicles, and proteins with neurons. This process helps remove harmful alpha-synuclein (a-syn) and tau aggregates, reducing oxidative stress and restoring normal gene expression. However, mutations in LRRK2 (Gly2019Ser) and TREM2 (T66M, R47H) impair TNT-mediated mitochondrial transfer, potentially exacerbating neurodegeneration.
These findings highlight the vital role of mitochondrial quality control and microglia-neuron interactions in neuroprotection. Targeting TNT-mediated mitochondrial transfer could offer new therapeutic strategies for neurodegenerative diseases.
For more details, read the full study: DOI: 10.1016/j.neuron.2024.06.029.
© Image Credits: Scheiblich et al. Neuron, 2024
The future of cancer immunotherapy will come through mitochondria: Cancer cells hijack T cells
- Details
- Published on 23 January 2025
Cancer cells ‘poison’ the immune system with tainted mitochondria
A study reveals how cancer cells suppress immune responses by transferring mutated mitochondria to tumor-infiltrating T cells. Researchers identified mitochondrial DNA mutations in T cells that matched mutations in the cancer cells they infiltrated. This transfer impaired T cell metabolism and function, leading to senescence and a diminished ability to fight tumors.
The transfer occurs via tunneling nanotubes and extracellular vesicles, with cancer-derived mitochondria resisting normal degradation processes. These dysfunctional mitochondria disrupt T cell energy production, leaving the immune cells unable to mount effective antitumor responses.
The findings could explain why some patients fail to respond to immune checkpoint inhibitors like PD-1 blockers. Targeting this mitochondrial transfer mechanism might open new doors for the future of cancer immunotherapy.
A dedicated workshop on mitochondrial transfer and its implications for cancer therapy will be organized at Targeting Mitochondria 2025.
For full Article: https://www.nature.com/articles/d41586-025-00176-2
Ketone Bodies: A New Approach to Brain Function and Neurodegenerative Diseases
- Details
- Published on 09 January 2025
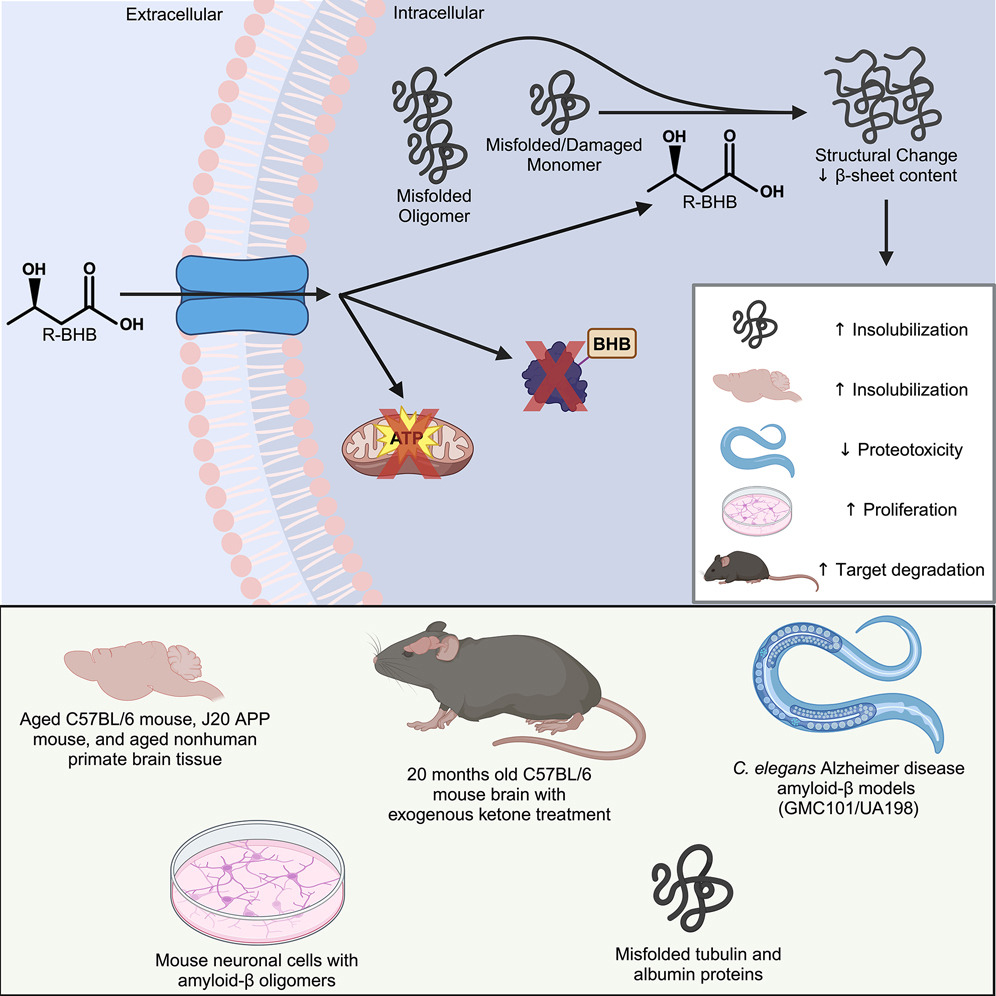
A recent study published in Cell Chemical Biology by researchers at the Buck Institute for Research on Aging has revealed intriguing new roles for ketone bodies, particularly β-hydroxybutyrate, in maintaining brain health. While ketone bodies are primarily recognized for their role in energy production, this research highlights their broader impact on mitochondrial function and protein homeostasis in the brain, offering promising insights for aging and neurodegenerative diseases like Alzheimer’s.
The study shows that β-hydroxybutyrate directly interacts with misfolded proteins in the brain, altering their structure and solubility to promote their clearance through autophagy. This process is crucial for supporting mitochondrial function, as it prevents the buildup of damaged proteins that can impair cellular health and energy production.
In addition, ketone bodies have a profound effect on protein quality control mechanisms within the brain. By influencing the proteome, ketones help enhance the clearance of dysfunctional proteins, ensuring that mitochondrial and cellular functions remain intact. This mechanism is vital for preserving the overall health of brain cells, especially as they age.
Experimental validation in animal models further supports these findings. When mice were fed ketone esters, the clearance of insoluble proteins was enhanced, preventing pathological aggregation. In nematodes expressing human amyloid beta, ketone treatment successfully reversed paralysis, suggesting a restoration of mitochondrial function and recovery from protein-induced damage.
These findings open up new potential therapeutic avenues for brain aging and neurodegenerative diseases. By manipulating ketone body levels, it may be possible to support mitochondrial function and facilitate the removal of damaged proteins from the brain. This approach could serve as a powerful strategy to mitigate the effects of aging on the brain and provide a novel way to treat neurodegenerative diseases.
The research underscores the emerging role of ketone bodies as signaling molecules that help regulate protein homeostasis and mitochondrial health in the brain. As mitochondrial dysfunction is a central factor in many neurodegenerative diseases, this study paves the way for new therapeutic approaches aimed at boosting mitochondrial resilience and improving brain health.
Image Credits: Sid Madhavan, Buck Institute for Research on Aging
Targeting Mitophagy in Neurodegenerative Diseases: A Promising Therapeutic Avenue
- Details
- Published on 15 January 2025
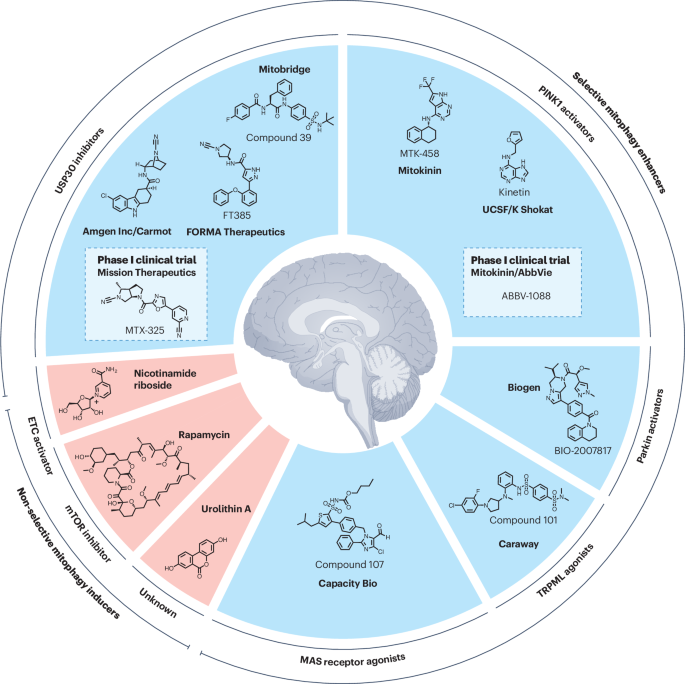
Mitochondrial dysfunction is a central feature of several neurodegenerative diseases, including Parkinson’s disease, amyotrophic lateral sclerosis (ALS), Alzheimer’s disease, and Huntington’s disease. A groundbreaking study published in Nature Reviews Drug Discovery by Odetta Antico, Paul W. Thompson, Nicholas T. Hertz, Miratul M. K. Muqit, and Laura E. Parton highlights the potential of targeting mitophagy — the cellular process that eliminates damaged mitochondria — as a therapeutic strategy for these debilitating conditions.
Mutations in genes related to mitophagy deficits are often linked to familial forms of Parkinson’s disease and ALS. Enhancing the mitophagy pathway could address this underlying pathogenic mechanism, providing neuroprotection and disease-modifying effects—an urgent unmet need in neurodegenerative disease management.
Excitingly, small molecules aimed at enhancing mitophagy, such as USP30 inhibitors and PINK1 activators, are now entering phase I clinical trials. This marks a pivotal moment in translating preclinical evidence into potential treatments that could revolutionize care for patients affected by these devastating diseases.
Figure Description: Selective and non-selective mitophagy activators.
Read the full article: Nature Reviews Drug Discovery (2025).
Pseudomonas Bacteria Disrupt Mitochondrial Energy Production to Evade Immune Response
- Details
- Published on 07 January 2025
New research reveals how Pseudomonas aeruginosa dampens macrophage bioenergetics by targeting mitochondrial pathways, opening doors for novel therapeutic approaches.
December 7, 2025
The bacterium Pseudomonas aeruginosa, commonly found in freshwater, hot tubs, and pools, poses a serious threat when it infects humans. Known for causing severe infections, particularly in immunocompromised individuals or burn patients, this opportunistic pathogen has evolved resistance to multiple antibiotics, making treatment increasingly difficult.
In a recent study published in eLife, Harvard researchers Laurence Rahme and Arijit Chakraborty uncovered a mechanism by which P. aeruginosa suppresses immune responses. They found that the bacterium produces a chemical, 2-aminoacetophenone (2-AA), that interferes with mitochondrial energy production in macrophages, crippling their ability to fight infections.
How 2-AA Targets Mitochondrial Energy Production
Macrophages, essential components of the innate immune system, rely heavily on energy generated by their mitochondria to engulf and eliminate pathogens. However, the presence of 2-AA significantly reduces adenosine triphosphate (ATP) levels in these cells, disrupting their bioenergetics.
ATP, the cell’s energy currency, is primarily produced through two pathways: glycolysis in the cytoplasm and oxidative phosphorylation in the mitochondria. While glycolysis yields a modest 2 ATP molecules per glucose, mitochondrial oxidative phosphorylation generates approximately 30 ATP molecules per glucose, making it the most efficient energy source.
Using Seahorse assay technology, the researchers measured oxygen consumption—a hallmark of oxidative phosphorylation—and observed a significant drop in cells exposed to 2-AA. This indicated that 2-AA specifically targets mitochondrial energy production, effectively shutting down the more efficient ATP-generating pathway.
Additionally, 2-AA was found to block pyruvate transport into mitochondria, leaving excess pyruvate in the cytoplasm. Pyruvate is a critical metabolite that fuels the Krebs cycle and oxidative phosphorylation within mitochondria. Without it, the energy output from macrophage mitochondria is severely impaired, leading to diminished immune responses.
Mitochondrial Dysfunction in Living Systems
To validate these findings, Rahme and her team conducted experiments in mice infected with either wild-type P. aeruginosa or a mutant strain unable to produce 2-AA. In mice infected with the wild-type bacteria, ATP levels in the spleen—a key organ for immune responses—dropped within 24 hours, while those infected with the mutant strain maintained normal energy levels.
Further analysis revealed reduced levels of acetyl-cofactor A, a mitochondrial metabolite derived from pyruvate, in wild-type infections. This confirmed that 2-AA disrupts mitochondrial bioenergetics. Importantly, the absence of 2-AA allowed macrophages to combat bacterial infections more effectively, leading to a lower bacterial burden by day 10.
Implications for Mitochondria-Targeted Therapies
As antibiotic resistance continues to rise, targeting host-pathogen interactions presents a promising therapeutic strategy. Kayeen Vadakkan, a microbiologist at St. Mary’s College, Thrissur, who was not involved in the study, highlighted the potential of targeting 2-AA to enhance mitochondrial function in macrophages. “We can complement our immune system,” Vadakkan said, suggesting that blocking 2-AA could restore macrophage energy production and improve their ability to fight infections.
Rahme’s laboratory is developing inhibitors of MvfR, a transcription factor necessary for 2-AA production. Early results show promise, but further studies are needed to evaluate their clinical safety and efficacy.
Beyond infection, 2-AA’s ability to suppress mitochondrial activity and reduce inflammation hints at its potential application in treating autoimmune diseases like rheumatoid arthritis and lupus, where overactive macrophages drive inflammation.12 “2-AA is a molecule which is anti-inflammatory in nature,” Chakraborty noted, emphasizing its dual therapeutic potential.
More Articles...
- Bridge Over Troubled Cells: Bone Marrow Stromal Cells Transfer Mitochondria to Boost T Cells
- Mitochondrial Dysfunction Disrupts Gut Microbiome, Possible Trigger for Crohn's Disease
- Study Shows Decreased Mitochondrial Creatine Kinase Impairs Muscle Function Independently of Insulin in Type 2 Diabetes
- Mitochondria’s Secret Strategy: How Cells Survive Starvation