Bridge Over Troubled Cells: Bone Marrow Stromal Cells Transfer Mitochondria to Boost T Cells
- Details
- Published on 16 December 2024
In a recent research published in Signal Transduction and Targeted Therapy, Lars Fabian Prinz,and his team present an innovative approach to combat T cell exhaustion using a bone marrow stromal cell (BMSC)-based mitochondrial transfer platform. This technique could revolutionize adoptive T cell therapies for cancer patients by addressing mitochondrial dysfunction in T cells.
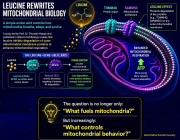
- Intercellular Mitochondrial Transfer: BMSCs transfer functional mitochondria to CD8+ T cells via tunneling nanotubes (TNTs), enhancing T cell metabolic fitness.
- Improved Anti-Tumor Activity: Transferred mitochondria significantly boosted T cell resistance to exhaustion and anti-tumor effectiveness, both in vitro and in vivo.
- Enhanced Therapeutic Potential: Mitochondrial-enriched (Mito+) T cells exhibited increased proliferation, reduced apoptosis, and higher cytotoxicity in tumor environments.
The study demonstrated enhanced outcomes in mouse models of melanoma and leukemia, with Mito+ T cells showing improved tumor suppression and survival rates. Challenges remain, including optimizing transfer efficiencies and overcoming tumor microenvironment constraints. However, this discovery opens doors for both cell-based and systemic therapies to modulate mitochondrial transfer for cancer treatment.
Figure Description
a Building on mitochondrial transfer techniques described in the literature, Baldwin and colleagues introduce a method to fortify CD8+ T cells with mitochondria transferred through tunneling nanotubules from bone marrow stromal cells (BMSCs). b The transfer results in T cells being more resistant against exhaustion and having higher anti-tumor activity in-vitro and in-vivo. c This could be applied to improve adoptive T cell therapies to treat patients with cancer. (Created with BioRender.com)
Source: https://www.nature.com/articles/s41392-024-02079-6
Image Credits: Prinz, L.F., Ullrich, R.T. & Chmielewski, M.M. B Sig Transduct Target Ther (2024).
Mitochondrial Dysfunction Disrupts Gut Microbiome, Possible Trigger for Crohn's Disease
- Details
- Published on 25 November 2024
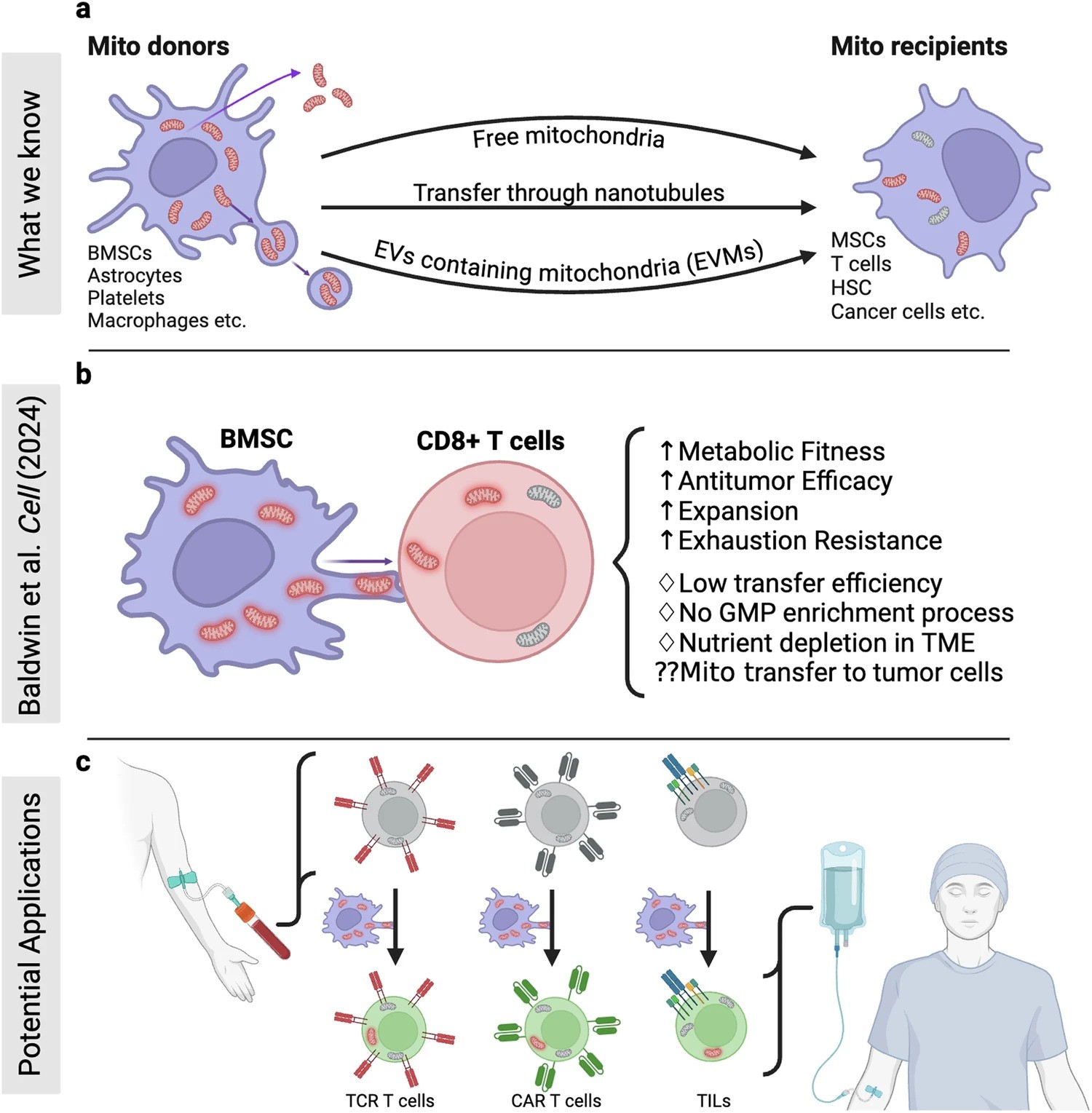
A recent study led by Prof. Dr. Dirk Haller at the Technical University of Munich (TUM) uncovers a critical connection between mitochondrial dysfunction and Crohn's disease (CD). The research demonstrates that defective mitochondria cause intestinal epithelial damage, triggering significant changes in the gut microbiome—key factors in the onset of this chronic inflammatory condition.
Key Findings:
- Mitochondrial dysfunction leads to tissue damage in the intestinal epithelium, mimicking Crohn's disease symptoms.
- Disruptions in mitochondrial function result in alterations to the gut microbiome composition.
- This marks the first demonstration of a causal link between mitochondrial health and gut inflammation.
Implications for Treatment: Current Crohn's disease therapies focus on symptom management, but these findings open the door to novel approaches that target mitochondrial repair, potentially offering more effective, long-term solutions for managing CD.
© Photo Credits: Urbauer, Elisabeth et al. Cell Host & Microbe (2024)
Mitochondria’s Secret Strategy: How Cells Survive Starvation
- Details
- Published on 18 November 2024

Mitochondria, often called the “powerhouses” of cells, are responsible for producing energy and vital molecules that cells need to function. However, until now, scientists have puzzled over how mitochondria manage to sustain these processes when cells are starved of nutrients. Researchers have uncovered that in low-resource conditions, mitochondria adopt a surprising strategy: they divide into two specialized forms. One form focuses on energy production to keep the cell powered, while the other concentrates on creating essential building blocks needed for repair and growth. This division of labor allows cells to survive and adapt even in challenging environments.
This remarkable discovery may also shed light on how some cancers thrive in hostile conditions within the body. Certain cancer cells appear to use the same mitochondrial strategy to fuel their growth and survival when nutrients are scarce. By splitting their functions, mitochondria in these cancer cells ensure the production of energy and key cellular components, enabling tumors to persist and grow even in environments that should inhibit them. Understanding this process offers new insights into cancer resilience and could pave the way for innovative treatments targeting mitochondrial functions in cancer cells.
This breakthrough highlights the extraordinary adaptability of mitochondria, offering a deeper understanding of cellular survival and disease mechanisms.
© World Mitochondria Society (WMS)
Study Shows Decreased Mitochondrial Creatine Kinase Impairs Muscle Function Independently of Insulin in Type 2 Diabetes
- Details
- Published on 25 November 2024
Published in Science Translational Medicine
Researchers from Karolinska Institutet have uncovered a key factor contributing to impaired muscle energy production in individuals with type 2 diabetes. The study reveals that people with type 2 diabetes have reduced levels of creatine kinase, a protein responsible for metabolizing and converting creatine in muscle cells. This deficiency hampers mitochondrial function—the "powerhouses" of cells—leading to decreased energy production and increased cellular stress.
Creatine, a compound naturally produced by the body and found in foods like meat and fish, is critical for muscle function. Although creatine supplementation is popular for enhancing exercise performance, elevated blood creatine levels have been linked to an increased risk of type 2 diabetes. This study demonstrates that the reduced levels of creatine kinase observed in people with type 2 diabetes lead to impaired creatine metabolism, explaining the accumulation of creatine in their bloodstream.
“Our findings suggest that impaired creatine metabolism is a consequence of type 2 diabetes rather than a cause,” says Professor Anna Krook from the Department of Physiology and Pharmacology at Karolinska Institutet, the study’s principal investigator.
Moreover, the study shows that reduced creatine kinase levels directly affect mitochondrial function, independent of creatine availability. This discovery highlights the multifaceted role of creatine kinase in cellular energy production.
“This is consistent with the poorer energy metabolism seen in people with type 2 diabetes,” adds Professor Krook. “In the future, regulating creatine kinase could be explored as a potential treatment strategy for metabolic diseases like obesity and diabetes.”
The next phase of research will focus on identifying the molecular mechanisms linking creatine kinase levels to mitochondrial function.
Military Medicine and Mitochondria: A Strong Interest in Targeted Strategies and Therapeutics
- Details
- Published on 27 September 2024

During the 15th Annual Meeting of the World Mitochondria Society (WMS), taking place in Berlin, Germany, groundbreaking research on mitochondria-targeted medicine in military settings will be highlighted. This research, particularly focused on Traumatic Brain Injury (TBI) and polytrauma, is of significant interest due to the high incidence of such injuries in combat, where multiple organs are affected by mechanical trauma, thermal injuries, or exposure to harmful agents such as chemical, biological, radiological, or nuclear materials.
Mitochondria-targeted medicine has emerged as a crucial area for military medicine, especially for addressing TBI. Research from the Walter Reed Army Institute of Research has emphasized the essential role of mitochondrial function in the acute phase following TBI. The research shows that TBI leads to bioenergetic failure and disruptions in calcium and redox balance, which are critical for recovery. The findings suggest that therapies aimed at restoring mitochondrial function may serve as an effective neuroprotective approach.
Moreover, the detection of mitochondria-specific markers in biofluids presents exciting opportunities for advancing diagnostics and therapeutics for TBI. These discoveries could lead to more accurate diagnoses and the development of personalized therapies that enhance recovery for military personnel.
 "The potential to target mitochondrial function in TBI treatment opens new doors for more effective interventions, offering hope for improved outcomes for service members affected by combat injuries", said Dr. Jignesh Pandya, Director of Brain Trauma Bioenergetics, Metabolism & Neurotherapeutics Development and leading researcher on the project.
"The potential to target mitochondrial function in TBI treatment opens new doors for more effective interventions, offering hope for improved outcomes for service members affected by combat injuries", said Dr. Jignesh Pandya, Director of Brain Trauma Bioenergetics, Metabolism & Neurotherapeutics Development and leading researcher on the project.
These findings, which underline the military's growing focus on mitochondria-centric therapeutic strategies, will be a focal point of discussion at the WMS Annual Meeting, attracting attention from the global scientific and medical community.
More about Dr. Pandya's talk at Targeting Mitochondria 2024.
About the World Mitochondria Society (WMS)
The World Mitochondria Society (WMS) is an international organization dedicated to advancing research on mitochondria and their role in health and disease. The society promotes global collaboration among scientists and medical professionals to accelerate breakthroughs in mitochondrial medicine, diagnostics, and therapies. The WMS Annual Meeting serves as a platform for presenting the latest scientific advancements and fostering dialogue on the future of mitochondrial research.
For more information on this research and its implications for military medicine, please contact the WMS (mitochondria[at]wms-site.com).
Speakers Line-up and other topics.
Abstracts to be presented
State of the Science Mitochondria Specific-Targets, Therapeutics and Biomarker Investigations following Traumatic Brain Injury in the US Military
Jignesh D. Pandya, Walter Reed Army Institute of Research, USA
Access Abstract
Variation in Mitochondrial Functions across Vital Organs and Brain Sub-Regions in a Swine Model: A Novel Reference Targets forTBI and Polytrauma
Anke H. Scultetus, Walter Reed Army Institute of Research, USA
Access Abstract