Nanoflowers help cells share mitochondria and restore energy
- Details
- Published on 10 February 2026
Scientists at Texas A&M University have developed an unusual tool to help damaged and aging cells recover their energy: microscopic particles called “nanoflowers.” These tiny structures can stimulate stem cells to produce and share mitochondria with neighboring cells, restoring cellular energy production.
Mitochondrial decline is a central feature of aging, neurodegeneration, and cardiovascular disease. When cells lose mitochondria, their ability to produce energy drops, leading to reduced function and increased vulnerability to stress.
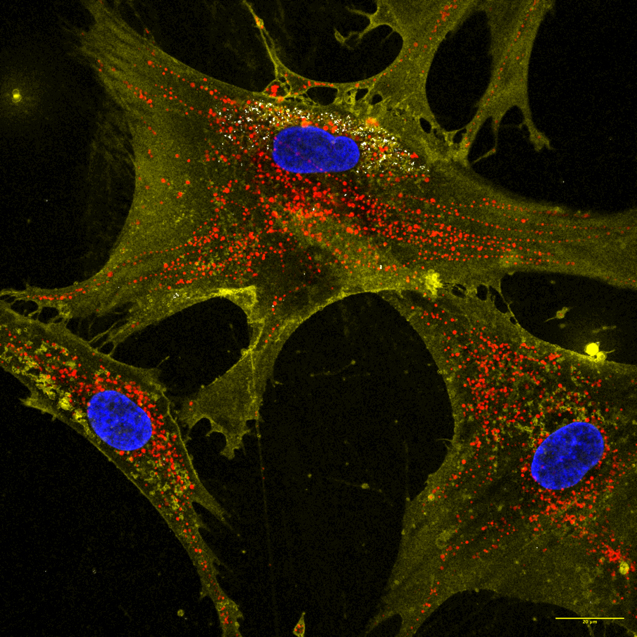
In this new study, researchers combined stem cells with flower-shaped nanoparticles made of molybdenum disulfide. The particles stimulated stem cells to produce about twice as many mitochondria as normal. These “mitochondrial bio-factory” cells then transferred the extra mitochondria to nearby damaged or aging cells.
The results were striking: recipient cells recovered energy production, improved survival, and resisted stress-induced cell death, including damage from chemotherapy like conditions.
While cells naturally exchange mitochondria at low levels, the nanoflower approach increased mitochondrial transfer by two- to four-fold, suggesting a way to amplify a natural repair mechanism rather than replace it.
Because the nanoparticles remain inside cells longer than conventional small molecule treatments, the approach could potentially enable long-lasting mitochondrial support therapies with less frequent dosing.
Although still at an early stage, the technology points toward a new regenerative strategy: boosting mitochondrial sharing between cells to restore tissue energy and resilience. Researchers suggest this approach could eventually be explored for conditions ranging from cardiomyopathy to neurodegenerative disease and muscle degeneration.
For the mitochondrial field, the study reinforces a growing idea: mitochondria are not only intracellular powerhouses, they are also transferable biological resources that can support tissue repair and cellular recovery.
About Image
Microscopic image showing how nanoflowers (white) help healthy cells (yellow) deliver energy-producing mitochondria (red) to neighboring cells. Nuclei are stained blue.
Courtesy Dr. Akhilesh K. Gaharwar.
Referance
Texas A&M University (2025). Texas A&M scientists use "nanoflowers" to recharge aging and damaged cells.
Read the full article
Cells share mitochondria to protect nerves from pain When cells share mitochondria, nerve pain can be reduced
- Details
- Published on 09 February 2026
A new study supported by the National Institutes of Health (NIH) shows that mitochondrial transfer between cells can protect nerves from pain-causing damage, revealing a promising therapeutic strategy for peripheral neuropathy.
Peripheral neuropathy often caused by chemotherapy or diabetes occurs when sensory neurons lose healthy mitochondria and can no longer produce enough energy to function properly. This leads to pain, weakness, and nerve dysfunction.
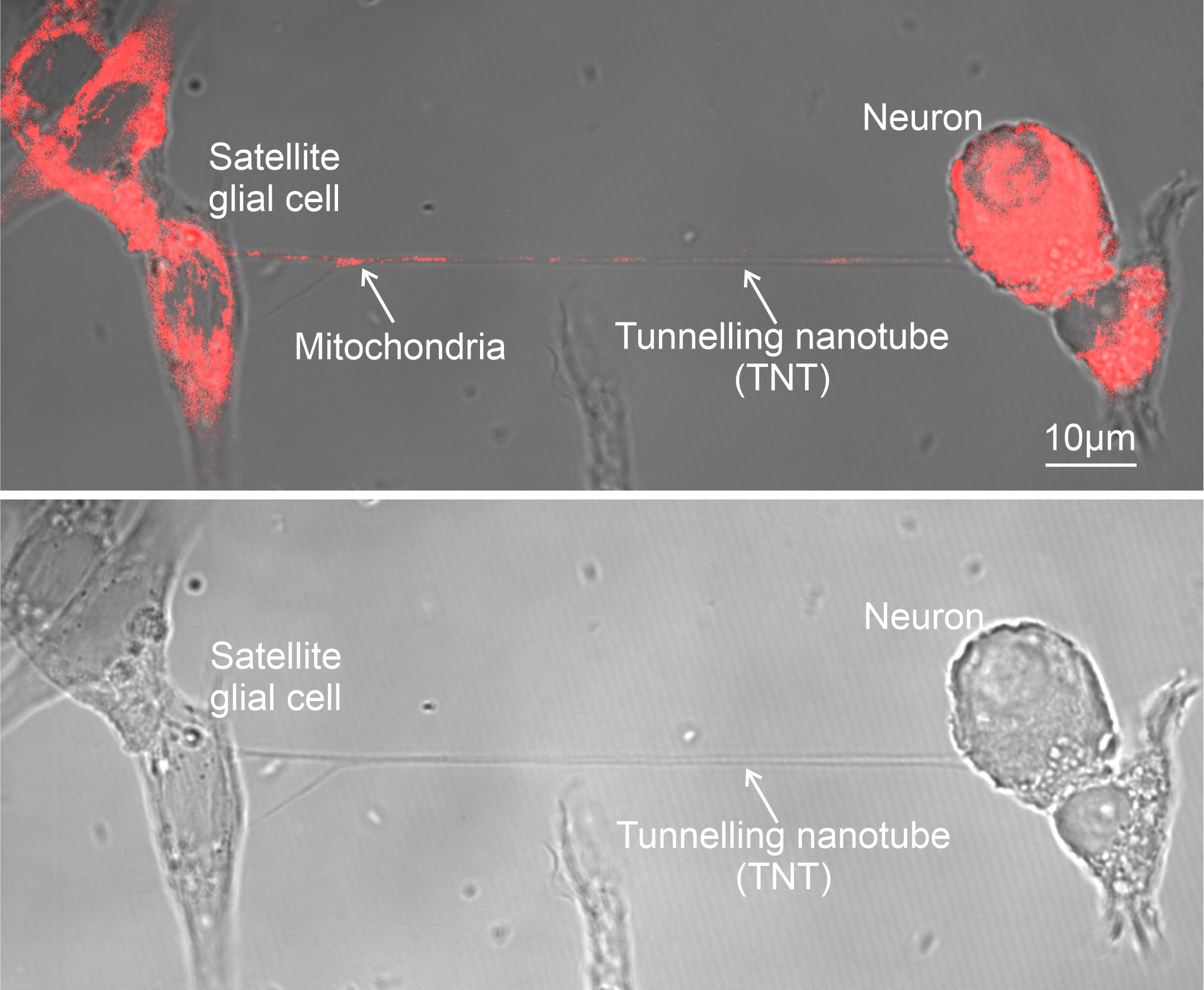
Researchers discovered that satellite glial cells, which surround sensory neurons in structures called dorsal root ganglia, can transfer mitochondria directly to neurons through tiny cellular bridges known as tunnelling nanotubes.
This mitochondrial transfer proved essential for nerve health. When scientists disrupted the formation of nanotubes or reduced levels of the motor protein MYO10, mitochondrial transfer declined and pain sensitivity increased in mice.
Encouragingly, restoring mitochondrial transfer reversed these effects. Injecting healthy satellite glial cells or even isolated mitochondria into affected nerve regions reduced pain sensitivity in mouse models of diabetic neuropathy and chemotherapy-induced nerve damage.
Human tissue samples showed similar biology: supporting cells from healthy donors displayed stronger mitochondrial-transfer capacity than those from people with diabetes.
Together, the findings highlight a growing concept in mitochondrial medicine: mitochondria can act as transferable therapeutic units, capable of restoring cellular energy and function in damaged tissues.
If translated clinically, strategies that enhance mitochondrial transfer or deliver healthy mitochondria could open a new path for treating chronic nerve pain and neurodegenerative conditions.
One of the hit topic of Targeting Mitochondria 2026 is Mitochondria Transfer and clinical impacts.
Image Credit
Scientists found that specialized cells, called satellite glial cells, can transfer mitochondria (red) to neurons through thin nanotubes that connect the two cells.
Ru-Rong Ji lab, Duke University School of Medicine
Referance
Mitochondrial transfer from glia to neurons protects against peripheral neuropathy. Xu J, Li Y, Novak C, Lee M, Yan Z, Bang S, McGinnis A, Chandra S, Zhang V, He W, Lechler T, Rodriguez Salazar MP, Eroglu C, Becker ML, Velmeshev D, Cheney RE, Ji RR. Nature. 2026 Jan 7. doi: 10.1038/s41586-025-09896-x. Epub ahead of print. PMID: 41501451.
Read the full article
Sugar first, mitochondria later: how brain immune cells respond to injury
- Details
- Published on 27 January 2026
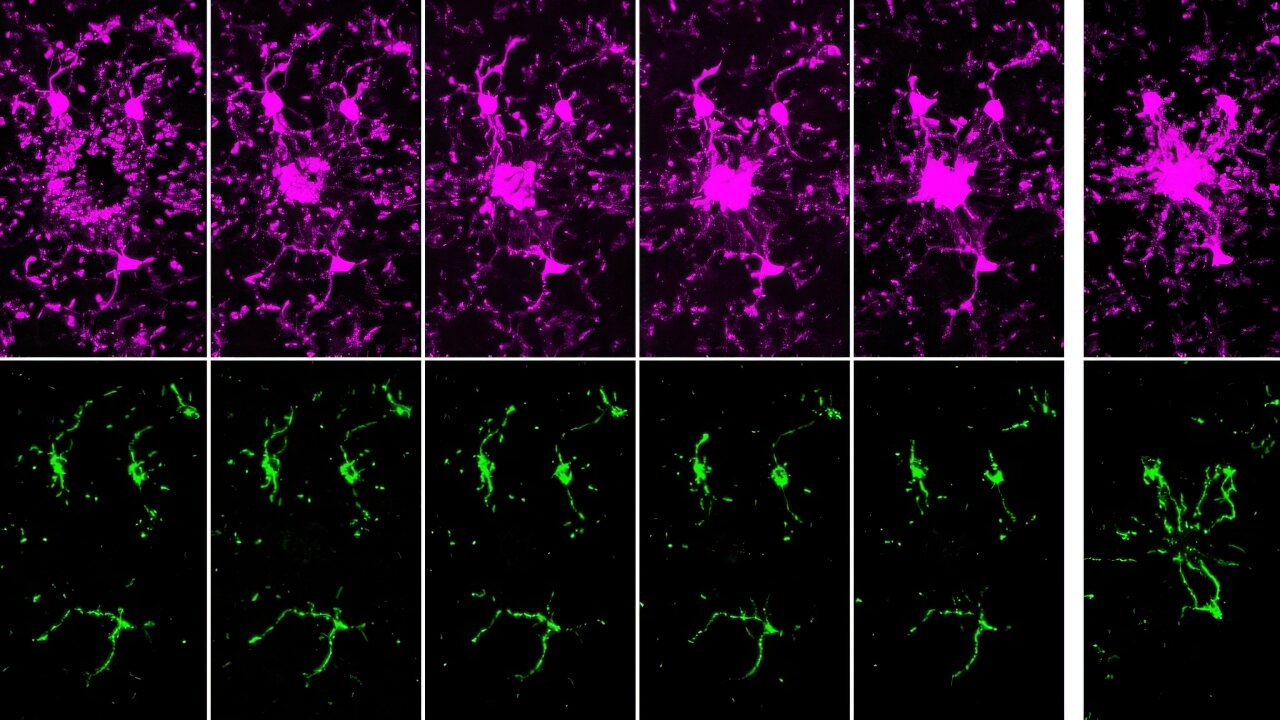
When the brain is injured, its immune cells don’t wait around. New research reveals that microglia, the brain’s resident immune cells, launch their first response using sugar-based energy, long before mitochondria enter the scene.
Using advanced live imaging, scientists observed that microglia rush toward damage and begin their work almost immediately even though their long cellular extensions contain few or no mitochondria at this early stage. Instead, these cells rely on glycolysis, a fast way to generate energy from sugar, allowing them to move quickly and react within minutes.
Only later do mitochondria arrive. Hours after the initial response, microglia reorganize their internal architecture, building microscopic transport routes that allow mitochondria to travel to the most active zones. Once there, mitochondria support sustained functions such as debris clearance and longer-term immune activity.
This two-step strategy challenges the traditional view of mitochondria as constant power suppliers. Instead, it shows that brain immune cells are metabolically flexible, choosing speed first and endurance second.
Why this matters: microglia play a central role in aging, neuroinflammation, and neurodegenerative diseases. Understanding how healthy microglia manage energy over time helps researchers identify what may go wrong in chronic brain disorders and how mitochondrial function might be restored or optimized.
In short, the brain’s immune response is not only fast it is smart, adaptive, and precisely timed.
The World Mitochondria Society will organize 2 meetings dedicated to mitochondria dynamics next April & October.
References
Pietramale, A.N., Bame, X., Doty, M.E. et al. Mitochondria are absent from microglial processes performing surveillance, chemotaxis, and phagocytic engulfment. Nat Commun16, 11104 (2025). https://doi.org/10.1038/s41467-025-66708-6
Read the full article.
A Mitochondrial Protein May Hold the Secret to Longevity
- Details
- Published on 05 February 2026
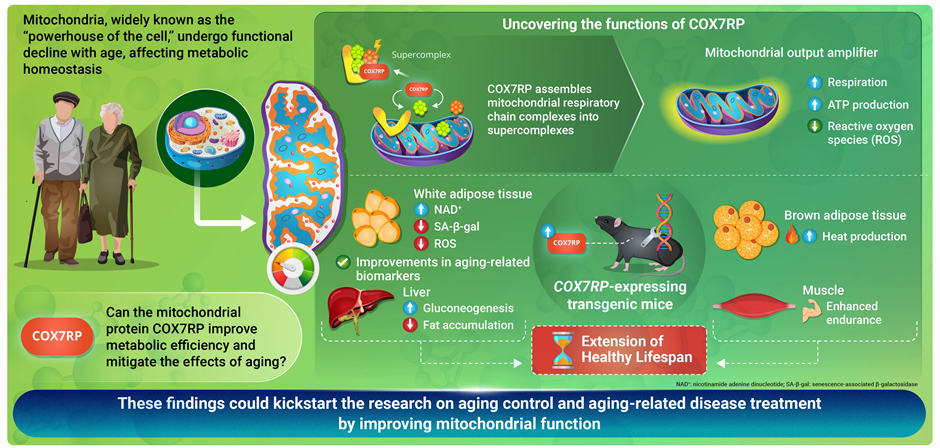
Researchers from the Tokyo Metropolitan Institute for Geriatrics and Gerontology have identified a mitochondrial protein that may play an important role in healthy aging. The study focuses on COX7RP, a protein involved in organizing mitochondrial respiratory supercomplexes, structures that improve cellular energy production efficiency.
Mitochondria are central to aging biology because they produce ATP, the energy required for cellular function. Declining mitochondrial performance is strongly associated with aging and age-related diseases. By increasing COX7RP levels in experimental models, researchers were able to improve mitochondrial efficiency, enhance metabolic health, and extend lifespan in mice.
The findings suggest that longevity may depend not only on metabolic activity itself, but on how efficiently mitochondrial systems are organized and coordinated. Respiratory supercomplexes appear to optimize electron transfer and reduce oxidative stress, helping cells maintain energy balance over time.
This work reinforces a growing idea in longevity science: aging is closely linked to the progressive decline of mitochondrial function and biological coordination. Improving mitochondrial organization and efficiency may therefore represent a promising strategy for extending healthspan.
The study also highlights how mitochondrial architecture, metabolism, and redox balance interact to influence aging trajectories, themes that resonate strongly with the systems-biology perspective explored at Targeting Longevity.
The World Mitochondria Society will organize 2 meetings dedicated to mitochondria dynamics next April & October.
Image Credit
Title: Exploring the link between COX7RP, a mitochondrial protein, and longevity
Caption: In a new study, researchers from Japan demonstrate that COX7RP, a mitochondrial protein, may play a key role in enhancing mitochondrial energy efficiency, leading not only to longer lifespans but also an extended "healthspan" via numerous health benefits.
Credit: Dr. Satoshi Inoue from Tokyo Metropolitan Institute for Geriatrics and Gerontology, Japan
Referance
Ikeda, K., S.Shiba, M.Yokoyama, et al. 2026. "Mitochondrial Respiratory Supercomplex Assembly Factor COX7RP Contributes to Lifespan Extension in Mice." Aging Cell25, no. 1: e70294. https://doi.org/10.1111/acel.70294.
Read the full article
Exercise May Help the Brain Heal After Stroke
- Details
- Published on 21 January 2026
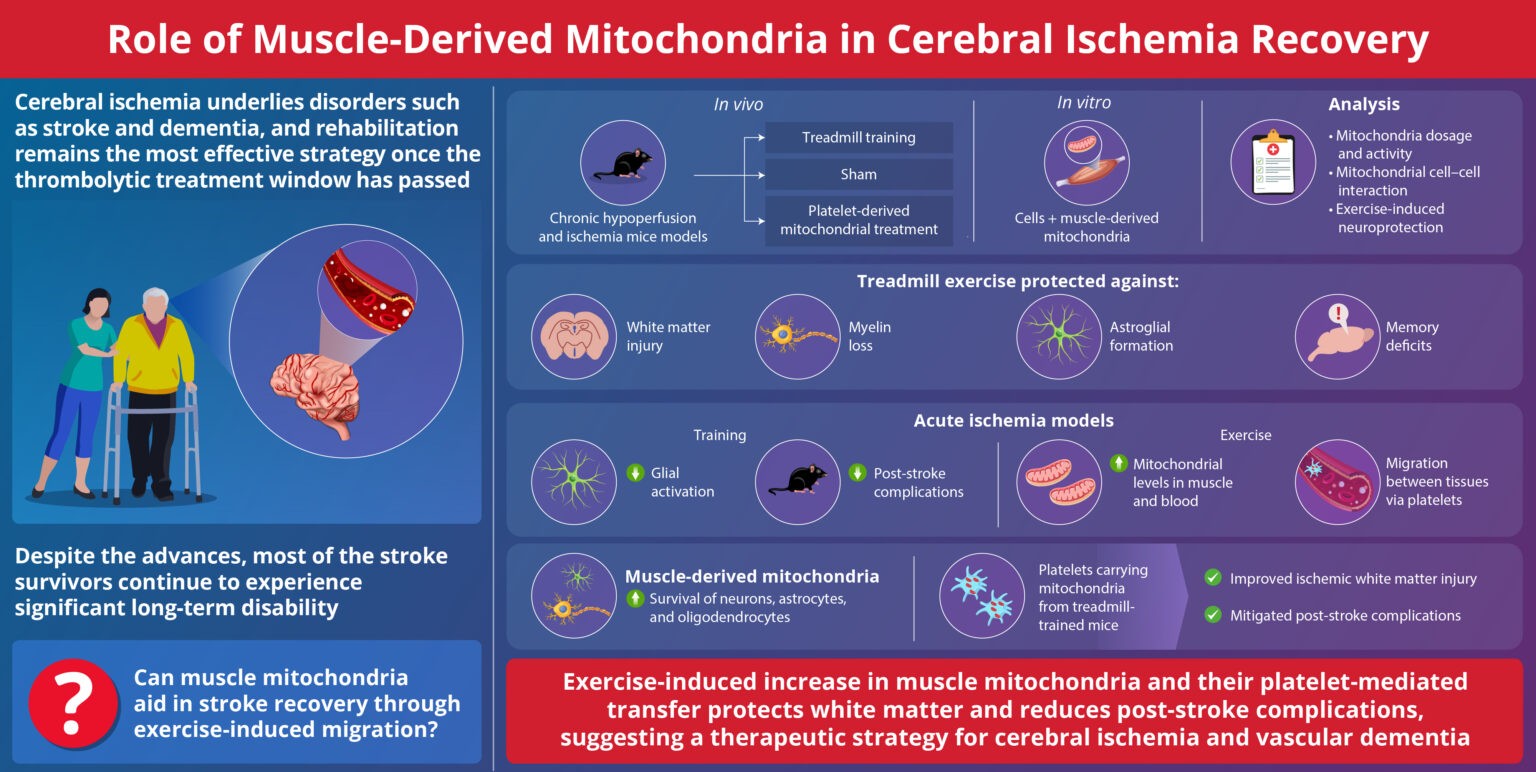
New research shows movement can send energy to damaged brain cells, opening new doors for therapy
Exercise has long been known to help people recover after stroke. Now scientists have found a possible reason why. A new study shows that physical activity may help the body deliver energy directly to injured parts of the brain, supporting repair and recovery.
The research suggests exercise is not only training muscles or improving balance. It may activate a natural healing process inside the body that helps brain cells survive and function after injury.
What they did
Researchers studied stroke in mice to closely observe what happens inside the body during recovery. They divided the animals into two groups. One group exercised regularly. The other group did not.
The scientists focused on mitochondria, the parts of cells that produce energy. They measured mitochondria in muscles, blood, and brain tissue.
They found that exercise increased the number of mitochondria in the blood. Platelets then carried these mitochondria through the bloodstream. After stroke, the mitochondria traveled into damaged areas of the brain and entered brain cells.
The researchers also tested movement, memory, and brain structure. Mice that exercised had less damage to brain tissue and performed better on recovery tests.
Why it matters
Stroke treatment options are limited. Emergency care can restore blood flow, but long term brain repair remains difficult. Rehabilitation depends largely on repeated physical practice.
This study adds a new layer of hope. It suggests exercise may work as a biological therapy, helping the brain at a cellular level, not only through practice and training.
Impact
The findings point to a new way of thinking about recovery. Exercise may act as a delivery system, sending energy from the body to the injured brain. This could help explain why movement improves outcomes even weeks or months after stroke.
The research also raises important questions. Could future therapies copy the benefits of exercise for patients who cannot move easily? Could similar approaches help people with dementia or other brain conditions?
Perspective
The study was done in animals, not humans, and more research is needed before clinical use. But the message is optimistic and grounded in biology.
The body is not passive after injury. With movement, it may activate its own repair tools.
Exercise may be more than rehabilitation. It may be part of the medicine itself.
These findings resonate strongly with the spirit of Targeting Mitochondria 2026, where dynamism is the central concept.
They remind us that health and recovery are not static processes, but living, energy-driven dialogues between organs, cells, and systems.
At TM2026, scientists, clinicians, and innovators will explore how movement, energy transfer, and mitochondrial intelligence can reshape our understanding of therapy from stroke recovery to neurodegeneration, aging, and resilience.
This study reinforces a simple but powerful idea: when biology moves, healing follows.
And sometimes, the most effective therapies begin not with drugs, but with restoring the natural dynamics of life itself.
References & Image Credits:
T.Inaba, N.Miyamoto, K.Hira, et al. “Mitochondrial Intercellular Transfer via Platelets After Physical Training Exerts Neuro-Glial Protection Against Cerebral Ischemia.” MedComm7, no. 2 (2026): e70590. https://doi.org/10.1002/mco2.70590
Image title: Exercise-induced mitochondria aid recovery from cerebral ischemia
Image caption: Researchers have demonstrated how mitochondria, which are abundant in muscle, could aid in stroke recovery through exercise-induced migration.
Image credit: Dr. Toshiki Inaba, Juntendo University School of Medicine, Japan
Read the full article
We are pleased to announce that the 17th Conference Targeting Mitochondria 2026 will be held in Berlin, Germany, from October 21-23. We look forward to welcoming you.