Pancreatic Cancer’s Hidden Weakness: Damaged Mitochondria May Be Its Achilles’ Heel
- Details
- Published on 04 May 2026

A simple mechanism, a powerful consequence: how mitochondrial damage fuels cancer and how to stop it.
A study published in Proceedings of the National Academy of Sciences, led by Erica Golemis and senior author Dario Altieri, identifies a new vulnerability in pancreatic cancer: damaged mitochondria can trigger inflammation that helps tumors survive.
When mitochondria send the wrong signal
The key player is Mic60, a protein that maintains mitochondrial structure. When Mic60 levels decrease, mitochondria become unstable and begin to leak double stranded RNA into the cell.
This leakage triggers a false alarm. The cell interprets it as a viral signal and activates inflammatory pathways, especially TLR3 and TRAF6. Instead of stopping cancer, this inflammation supports tumor growth and survival.
A vulnerability inside the tumor
Pancreatic cancer cells appear to depend on this mitochondrial driven inflammation. When researchers blocked this pathway in preclinical models, tumor growth was reduced, while normal cells were less affected.
This suggests a clear therapeutic opportunity: targeting a mechanism the tumor relies on.
Next steps toward potential therapies
Researchers are now focusing on two priorities:
Understanding how Mic60 damages mitochondrial membranes, allowing double stranded RNA to escape and initiate inflammation
Developing and evaluating TLR3 TRAF6 inhibitors as a potential new treatment approach
WMS Perspective
This study reinforces a key message: mitochondria are not passive. They actively drive inflammation and disease.
Strategic question
Can mitochondrial dysfunction be transformed into a precise therapeutic target in cancer?
This research suggests it can.
Can Mitochondria Create New Organelles? A Discovery Reopens a Major Question in Cell Biology
- Details
- Published on 28 April 2026
- Researchers are discovering that mitochondria (blue), known as cells' power plants, also play parts in cells' immune responses.Credit: David M. Phillips/Science Photo Library
A new report in Nature, written by Viviane Callier, describes a provocative discovery: when the parasite Toxoplasma gondii invades host cells, mitochondria respond by shedding their outer layers to form new cellular compartments that can digest molecular waste. The article reports on a 2026 bioRxiv preprint by Lena Pernas, Xin Li, Yue Sun, and José M. Delgado.
This observation goes far beyond classical mitochondrial dynamics.
Mitochondria are usually presented as energy-producing organelles. They are also recognized as regulators of immunity, redox signaling, stress adaptation, and cell death. This new work adds a more radical possibility: mitochondria may actively participate in building new cellular architecture.
The discovery raises three strategic questions for the field.
First, mitochondrial immunity.
Mitochondria do not simply suffer during infection. They sense danger, reorganize their membranes, communicate with immune pathways, and may now be viewed as contributors to the formation of defense-associated compartments.
Second, organelle evolution.
The finding supports a broader evolutionary hypothesis: ancient mitochondria may have helped shape the internal compartmentalization of modern eukaryotic cells. In other words, mitochondria may not only be descendants of bacteria. They may also have been architects of cellular complexity.
Third, mitochondrial intelligence.
This discovery reinforces one of the central messages of Targeting Mitochondria 2026: mitochondria are not passive power stations. They are adaptive, communicative, and structurally dynamic organelles able to influence cell fate under stress.
This topic strongly fits the spirit of Targeting Mitochondria 2026, where the World Mitochondria Society will explore mitochondria as decision-making hubs in immunity, infection, organelle communication, aging, and disease vulnerability.
Key question:
Are mitochondria only organelles, or can they also generate new cellular organization?
Red Blood Cells Lose Their Mitochondria: But Not Before Building the Machinery of Oxygen Transport
- Details
- Published on 24 April 2026
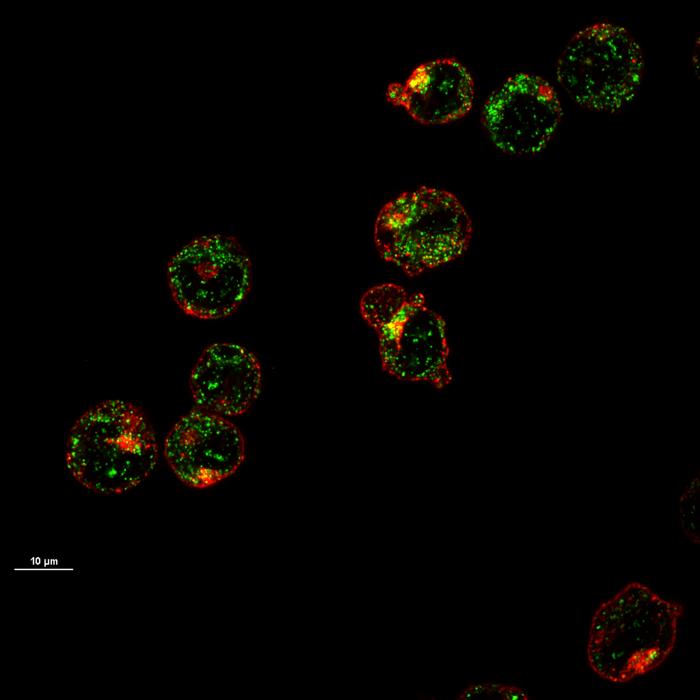
- Maturing precursor red blood cells display Heme Responsive Gene 1(HRG1) transporters (in green). These protein complexes are expressed on cell membranes and shuttle the iron-containing molecule heme from outside to the interior of the cell.
Red Blood Cells Lose Their Mitochondria: But Not Before Building the Machinery of Oxygen Transport
A new study reveals how developing red blood cells import heme to complete oxygen-carrying capacity under stress.
A new study from the University of Maryland School of Medicine led by Iqbal Hamza, published in Science, reveals a surprising mechanism that helps young red blood cells produce hemoglobin when the body is under stress.
Red blood cells carry oxygen through the body using hemoglobin, the red protein that captures oxygen in the lungs and delivers it to tissues. To build hemoglobin, developing red blood cells need heme, an iron-containing molecule that allows oxygen to bind.
Mature red blood cells do not have mitochondria. They lose their mitochondria during development to become highly specialized oxygen carriers. But before they mature, young red blood cells still contain mitochondria, and these mitochondria help produce heme.
This creates an important biological question: How can red blood cells continue building enough hemoglobin while they are losing the mitochondria normally involved in heme production?
The new study identifies a backup pathway. Researchers showed that immature red blood cells, called erythroblasts, can import heme from surrounding cells through a transporter called HRG1. This external heme supply becomes especially important when the body must rapidly produce new red blood cells, such as after blood loss, during anemia, or under low oxygen conditions.
In mouse models, when HRG1 was removed, developing red blood cells could not accumulate enough hemoglobin, failed to mature properly, and the animals became anemic under stress. The researchers also found that modifying HRG1 activity improved red blood cell production in a model of β-thalassemia, an inherited disorder affecting hemoglobin formation.
For mitochondrial biology, this discovery is important because it highlights a transition phase in red blood cell development. Mitochondria help young blood cells produce heme, but as these cells mature and remove their mitochondria, they may rely on heme supplied from outside the cell to complete hemoglobin production.
This finding changes the way we understand blood formation. Hemoglobin production is not only an internal process driven by mitochondria. It also depends on communication and material exchange between cells.
The discovery opens new perspectives for anemia, β-thalassemia, sickle cell disease, and other conditions where heme balance, oxidative stress, and red blood cell production are disrupted.
It also reinforces a broader concept central to mitochondrial medicine: mitochondria are not only energy producers. They participate in the construction, adaptation, and resilience of cells during stress.
Take-home message
Young red blood cells use mitochondria to help make heme. Mature red blood cells no longer have mitochondria. This study shows that, during the transition, developing blood cells can import heme from nearby cells through HRG1 to finish building hemoglobin, especially when the body is under stress.
Read the complete news here: www.eurekalert.org
Article: Audrey Belot et al., A cell-nonautonomous heme acquisition pathway enables erythroid hemoglobinization under stress.Science392, 388-394(2026). DOI:10.1126/science.aea0552
Mitochondria Help Control Skin Pigmentation: A New Organelle Dialogue Revealed
- Details
- Published on 27 April 2026
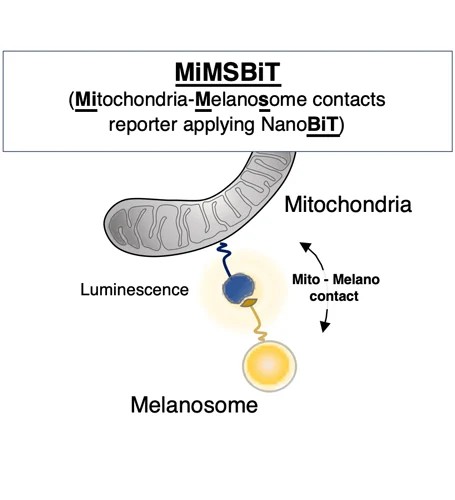
- Schematic overview of MiMSBiT, a reversible system for quantifying mitochondria–melanosome contacts in living cells.
A new study by Shigeru Yanagi, Isshin Shiiba and colleagues from the University of Tokyo shows that mitochondria do more than produce energy: they physically communicate with melanosomes, the organelles responsible for producing and storing melanin.
Researchers developed a new live-cell tool called MiMSBiT, allowing them to measure contacts between mitochondria and melanosomes in real time. Using this approach, they found that mitochondria directly interact with melanosomes and provide ATP locally, supporting melanosome maturation and melanin production.
The study, published in Nature Communications, identifies a molecular bridge involving STIM1 on melanosomes and Mitofusin 2 / MFN2 on mitochondria. When this contact is disrupted, melanosome maturation is impaired and melanin synthesis decreases.
This discovery adds a new dimension to pigmentation biology. Skin color and pigment changes are not only controlled by enzymes and hormones; they also depend on organelle communication inside the cell. Mitochondria appear to act as local energy and signaling partners, helping melanosomes reach the correct stage of maturation.
The implications may extend beyond basic pigmentation. The authors suggest that these findings could support future research on aging-related pigment changes and pigmentation disorders.
Impact on mitochondrial medicine
This study reinforces a central message of modern mitochondrial biology: mitochondria are not isolated power plants. They are communication hubs, interacting with other organelles to regulate cell identity, adaptation, and function.
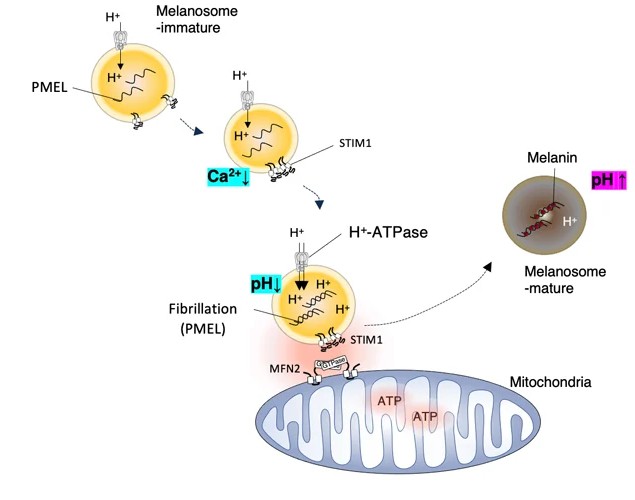
Mfn2 on mitochondria and STIM1 on melanosomes mediate mitochondria–melanosome contact, promoting melanosome maturation and facilitating melanin pigmentation.
At Targeting Mitochondria 2026, this type of discovery will be central to discussions on mitochondrial intelligence, organelle communication, aging, and new therapeutic strategies. This topic will be discuss also Skin Challenge 2026.
Read the complete news by Shiiba, I., Ishikawa, Y., Oshio, H. et al. STIM1-Mitofusin2 interactions tether mitochondria and melanosome contacts that promote melanosome maturation. Nat Commun 17, 3593 (2026). here
“MitoCatch” delivers healthy mitochondria to diseased cells
- Details
- Published on 24 April 2026
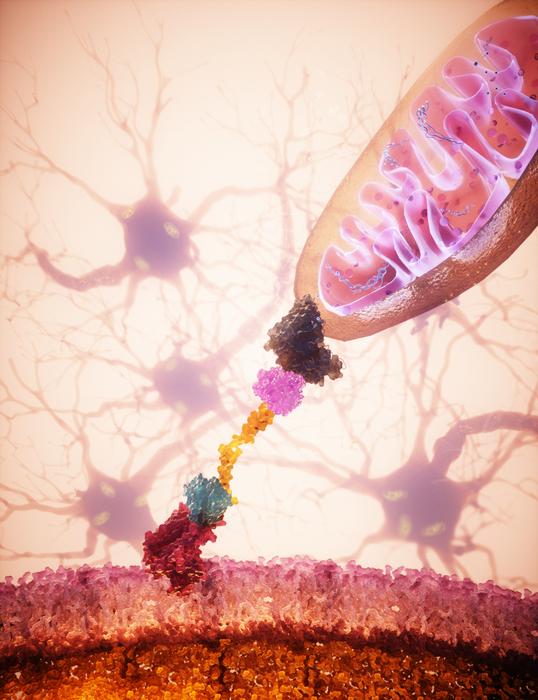
- Extracellular donor mitochondria are delivered specifically
to neurons via bispecific protein binder
New technology opens therapeutic opportunities for neurodegenerative diseases, optic nerve disorders, and heart failure.
Scientists led by Botond Roska at the Institute of Molecular and Clinical Ophthalmology Basel (IOB) have developed MitoCatch, a groundbreaking system that enables targeted delivery of healthy mitochondria to specific cell types affected by disease. This innovation is a major step toward precision mitochondrial therapy.
Mitochondrial dysfunction contributes to numerous currently untreatable conditions, including neurodegenerative disorders such as Parkinson’s and Alzheimer’s, optic nerve atrophy, and heart failure. While transplantation of healthy mitochondria has been explored as a potential therapy, conventional methods lack the specificity and efficiency needed to reach disease-affected cells.
MitoCatch overcomes these limitations by using engineered protein binders to link donor mitochondria to target cells. The system employs three complementary strategies: binders on the cell surface (MitoCatch-C), binders on mitochondria (MitoCatch-M), and bispecific binders bridging mitochondria and cell membranes (MitoCatch-Bi). By tuning binder affinity and multivalent interactions, researchers achieved efficient, cell type-specific mitochondrial delivery in human and mouse models across multiple cell types.
Key findings from the work of first authors Temurkhan Ayupov and Veronica Moreno-Juan and collaborators:
- MitoCatch directs donor mitochondria to specific cell types, including neurons, retinal, cardiac, endothelial, and immune cells.
- Binder-mediated mitochondrial delivery increases uptake compared to untargeted methods.
- Donor mitochondria internalized via MitoCatch become cytosol-exposed, are dynamic, and capable of fusion / fission within recipient cells.
- Protein binders can be engineered to tune delivery efficiency and specificity.
- Targeted transplantation improved survival of damaged neurons in vitro and retinal ganglion cells in vivo, and was well tolerated in animal models, with no detectable immune response.
MitoCatch enables the first efficient, targeted delivery of healthy mitochondria to the cells most affected by disease. By overcoming the long-standing challenge of cell type-specific mitochondrial transplantation, MitoCatch opens new avenues for research and potential therapies for multiple diseases linked to mitochondrial dysfunction. Its versatility across multiple human cell types positions it as a pioneering platform for precision mitochondrial medicine.
News source: www.eurekalert.org
Article: Ayupov, T., Moreno-Juan, V., Curtoni, S. et al. Cell-type-targeted mitochondrial transplantation rescues cell degeneration. Nature (2026). https://doi.org/10.1038/s41586-026-10391-0
Image © IOB, 2026
More Articles...
- How mitochondria organize their DNA to sustain cellular energy
- Mitochondria Could Help Make Cancer Immunotherapy More Effective
- Organelle Therapy Encapsulated Mitochondria Restore Function in Parkinson’s and Genetic Disorders: A Breakthrough
- Could fixing mitochondria restore insulin-producing cells?