Regulatory Role of Extracellular Vesicles on Mitochondria Under Hypoxia
- Details
- Published on 10 March 2022
News Release, World Mitochondria Society, Berlin - Germany – March 10, 2022
Mitochondria are indispensable organelles for maintaining cell energy metabolism, and also are necessary to retain cell biological function by transmitting information as signal organelles. Hypoxia, one of the important cellular stresses, can directly regulates mitochondrial metabolites and mitochondrial reactive oxygen species (mROS), which affects the nuclear gene expression through mitochondrial retrograde signal pathways, and also promotes the delivery of signal components into cytoplasm, causing cellular injury. In addition, mitochondria can also trigger adaptive mechanisms to maintain mitochondrial function in response to hypoxia.
Extracellular vesicles (EVs), as a medium of information transmission between cells, can change the biological effects of receptor cells by the release of cargo:
- Nucleic acids
- Proteins
- Lipids
- Mitochondria and their compositions
The secretion of EVs increases in cells under hypoxia, which indirectly changes the mitochondrial function through the uptake of contents by the receptor cells.
This article focuses on the mitochondrial regulation indirectly through EVs under hypoxia, and the possible mechanisms that EVs cause the changes in mitochondrial function. It also discusses the significance of this EV-mitochondria axis in hypoxic diseases.
You will have the chance to know more about extracellular vesicle-mitochondrial relationships and many more mitochondria related topics in Targeting Mitochondria 2022.
Dr. Marc Germain and Dr. Devika S. Manickam will both join us in October to cover this EV-mitochondria relationship, so don't forget to reserve your spot!
Media contact:
World Mitochondria Society
This email address is being protected from spambots. You need JavaScript enabled to view it.
+33-1-5504-7755
Targeting Mitochondria 2022 Congress
October 26-28, 2022 - Berlin, Germany
wms-site.com
Mitochondria Rich Extracellular Vesicles & Ageing: What is the Link?
- Details
- Published on 10 March 2022
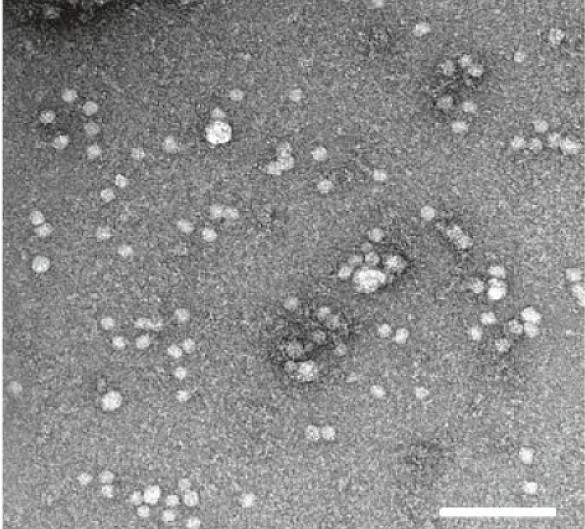
Electron microscopy showed EVs of typical size and morphology
News Release, Wolrd Mitochondria Society, Berlin - Germany – March 10, 2022
The accumulation of oxidative damage to mitochondria and mitochondrial DNA (mtDNA) plays a major role in aging, "the mitochondrial free radical theory of aging". Circulating cell-free mtDNA (ccf-mtDNA) in blood can be considered as disease biomarkers.
Extracellular vesicles (EVs), the small lipid-bound vesicles capable of shuttling cargo, take part in intercellular communication systems.
In thier fascinating research, Lazo et al.:
- Reported that a portion of ccf-mtDNA in plasma is encapsulated in EVs.
- Studied whether EV mtDNA levels vary upon aging by analyzing mtDNA in EVs from individuals aged 30–64 years cross-sectionally and longitudinally.
They reported the following:
- EV mtDNA levels decreased with age.
- The maximal mitochondrial respiration of cultured cells was differentially affected by EVs from old and young donors.
In conclusion, their results suggested that plasma mtDNA is present in EVs, that the level of EV-derived mtDNA is associated with age, and that EVs affect mitochondrial energetics in an EV age-dependent manner.
In Targeting Mitochondria 2022, a whole session will be dedicated to Extracellular Vesicles & Mitochondria: The Target, in which Dr. Marc Germain and Dr. Devika S. Manickam will share with us their
Media contact:
World Mitochondria Society
This email address is being protected from spambots. You need JavaScript enabled to view it.
+33-1-5504-7755
Targeting Mitochondria 2022 Congress
October 26-28, 2022 - Berlin, Germany
wms-site.com
Mitochondrial Base Editor: A New Strategy to Target Mitochondria Dysfunctions
- Details
- Published on 22 February 2022
Scientists at the University of Cambridge have shown that it is possible to modify the mitochondrial genome in live mice, paving the way for new treatments for incurable mitochondrial disorders.

News Release, World Mitochondria Society, Berlin - Germany – February 22, 2022
Mitochondrial DNA makes up only 0.1% of the overall human genome and is passed down exclusively from mother to child. Faults in our mitochondrial DNA can affect how well the mitochondria operate, leading to mitochondrial diseases, serious and often fatal conditions that affect around 1 in 5,000 people.
There are typically around 1,000 copies of mitochondrial DNA in each cell, and the percentage of these that are damaged, or mutated, will determine whether a person will suffer from mitochondrial disease or not. Usually, more than 60% of the mitochondria in a cell need to be faulty for the disease to emerge, and the more defective mitochondria a person has, the more severe their disease will be. If the percentage of defective DNA could be reduced, the disease could potentially be treated.
Dr. Minczuk and colleagues used a biological tool known as a mitochondrial base editor to edit the mitochondrial DNA of live mice. The treatment is delivered into the bloodstream of the mouse using a modified virus, which is then taken up by its cells. The tool looks for a unique sequence of base pairs, combinations of the A, C, G and T molecules that make up DNA. It then changes the DNA base, in this case, changing a C to a T. This would, in principle, enable the tool to correct certain 'spelling mistakes' that cause the mitochondria to malfunction.
There are currently no suitable mouse models of mitochondrial DNA diseases, so the researchers used healthy mice to test the mitochondrial base editors. However, it shows that it is possible to edit mitochondrial DNA genes in a live animal.
You will have the chance to know more about the potential of this mitochondrial base editor in treating mitochondrial disorder in Targeting Mitochondria 2022 with Dr. Pedro Pinheiro, from the University of Cambridge - United Kingdom.
Read more about In-vivo Mitochondrial Base Editing.
© Image - motoviurii - fr.freepik.com
Media contact:
World Mitochondria Society
This email address is being protected from spambots. You need JavaScript enabled to view it.
+33-1-5504-7755
Targeting Mitochondria 2022 Congress
October 26-28, 2022 - Berlin, Germany
wms-site.com
Mitochondria As Microlenses: Strategic Role In Eyesight
- Details
- Published on 07 March 2022
News Release, World Mitochondria Society, Berlin - Germany – March 7, 2022
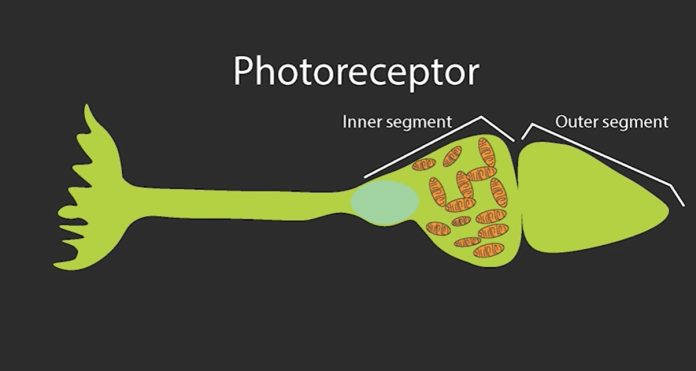
A study by Researchers at the National Eye Institute (NEI) on ground squirrels shows a fascinating phenomenon that mitochondria appear to have a dual purpose: their well-established metabolic role producing energy, as well as this optical effect. This was revealed by their act as micro-lenses that redirect light to the tapering outer reaches of these cells where light is converted into electrical signals.
Once light reaches the retina, it must pass through several neural layers to reach the outer segment of photoreceptors, where light’s physical energy is converted into neural signals through a process called phototransduction. Between the inner and outer segment of the cone photoreceptors lie a dense bundle of mitochondria that light must traverse to be transduced. Although it might appear these mitochondria pose an obstacle to the process of vision by either scattering or absorbing light, the current study shows they serve a unique function to facilitate vision.
Those bundles of mitochondria would seem to work against the process of vision either by scattering light or absorbing it. So, Li’s team set out to investigate their purpose by studying cone photoreceptors from the 13-lined ground squirrel.
Using a modified confocal microscope to observe the optical properties of living cone mitochondria exposed to light, the researchers observed that instead of scattering light, the tightly packed mitochondria concentrated light along a pencil-like trajectory onto the light-sensitive outer segment. High-resolution mitochondrial reconstructions corroborated the live-imaging findings. In addition, the authors show remodeling mitochondrial architecture affects this concentration of light.
In this study, Li found that the lens-like effect of mitochondria followed a similar directional light intensity profile. That is, depending on light source location, the mitochondria focused light into the outer segment of the cell along trajectories that mirrored those observed from the Stiles-Crawford effect.
The study also sheds new light on how our eyes may have evolved. Within the photoreceptors of birds and reptiles, tiny oil droplets at the junction of the inner and outer segments that may play an optical role are reminiscent of the lipid-rich mitochondria of cones in the current study on ground squirrels. Moreover, the mitochondrial “microlens” in mammalian cone photoreceptors is functionally like the biological effect achieved by the compound eye in insects.
“This insight conceptually bridges compound eyes in arthropods with the camera eyes of vertebrates, two independently evolved image-forming systems, demonstrating the power of convergent evolution,” Li said.
More interesting studies on the hidden functions of mitochondria will be introduced in Targeting Mitochondria 2022.
Read more about the micro-lense function of mitochondria.
Media contact:
World Mitochondria Society
This email address is being protected from spambots. You need JavaScript enabled to view it.
+33-1-5504-7755
Targeting Mitochondria 2022 Congress
October 26-28, 2022 - Berlin, Germany
wms-site.com
Nerve Cells Damage Could be Reversed Using Peptide
- Details
- Published on 22 February 2022

News Release, Wolrd Mitochondria Society, Berlin - Germany – February 22, 2022
Jun Li et. al research at the University of Illinois Chicago presented promising results for a treatment to stop nerve cell degeneration that happens in some types of disorders, such as hereditary spastic paraplegia and Parkinson's disease, which can cause significant disability.
The reseach team was able to use human cells that they transformed into stem cells and then modified to become nerve cells with the genetic disorder for a particular type of hereditary spastic paraplegia.The study looked at how the long axons that carry messages between nerve cells in the brain can break down, which causes increasingly worse tightening of the leg muscles, leading to imbalance and eventually paralysis, in addition to other symptoms.
Jun Li says: "What we found was that the mitochondria in these cells were breaking apart, what we call mitochondrial fission, and that caused the axons to be shorter and less effective at carrying messages to the brain". Also, "We then looked at whether a particular agent would change the way the nerve cells function - and it did. It inhibited the mitochondrial fission and let the nerve cells grow normally and also stopped further damage."
In conclusion, this agent (a particular chain of amino acids called a peptide) could prove to be useful for a drug or other therapy to stop the nerve cells from becoming damaged or reverse the course of the damage. Moreover, using gene therapy could prevent mitochondrial damage, providing another strategy to reverse the nerve damage.
Similar interesting researches will be discussed in detail in the 13th World Annual Meeting of WMS on Targeting Mitochondria which will be held on October 2022 in Berlin.
Photo Credit: rawpixel.com - www.freepik.com
Media contact:
World Mitochondria Society
This email address is being protected from spambots. You need JavaScript enabled to view it.
+33-1-5504-7755
Targeting Mitochondria 2022 Congress
October 26-28, 2022 - Berlin, Germany
wms-site.com