A new driver of inflammation in aging: Mitochondrial RNA leakage
- Details
- Published on 14 January 2026
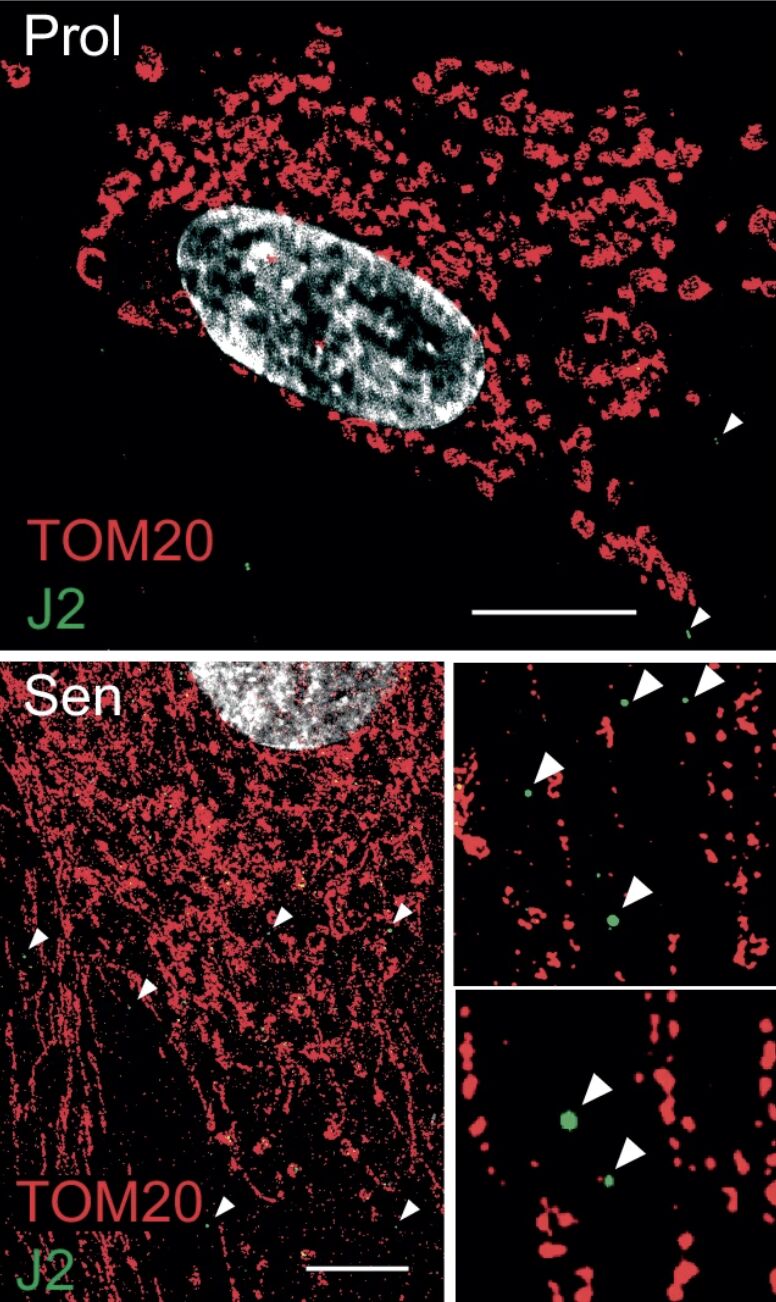
A major new study authored by Stella Victorelli and Madeline Eppard and led by Joao Passos, of Mayo Clinic, uncovers a previously unknown mechanism driving chronic inflammation in aging.
The research indicates that mitochondrial RNA (mtRNA) leaking into the cytosol of senescent cells acts as a key trigger for the senescence-associated secretory phenotype (SASP), a significant factor contributing to tissue dysfunction and age-related diseases.
What the study found:
- Senescent cells accumulate mtRNA in the cytosol, where it activates innate immune RNA sensors RIG-I and MDA5, leading to Mitochondrial Antiviral Signaling protein (MAVS) aggregation and inflammatory signaling.
- The study identifies BAX and BAK-dependent mitochondrial membrane permeabilization as the mechanism allowing mtRNA leakage. Genetic deletion of BAX/BAK suppresses SASP both in vitro and in vivo.
- In a mouse model of metabolic dysfunction–associated steatohepatitis (MASH), inhibiting this mtRNA-RNA sensing axis reduced liver inflammation and fibrosis markers, demonstrating physiological relevance.
Why it matters:
This work expands the aging paradigm beyond mitochondrial DNA by identifying mtRNA as a potent endogenous danger signal. It reveals a new mitochondria-to-cytosol signaling axis that fuels chronic inflammation in aging and age-related disease. It highlights RNA sensing, MAVS, and mitochondrial permeability as promising therapeutic targets to mitigate inflammaging without eliminating senescent cells.
References:
Victorelli, S., Eppard, M., Martini, H. et al. Mitochondrial RNA cytosolic leakage drives the SASP. Nat Commun16, 10992 (2025). https://doi.org/10.1038/s41467-025-66159-z